Browse articles on EyeWorld.org from the Cornea section. EyeWorld is the award-winning member publication of ASCRS. The magazine provides news and updates from the Society as well as clinical features in the areas of cataract, refractive, cornea, glaucoma, and practice management. It publishes quarterly with editorial direction from its medical editorial board and staff.
For about 7 years, there has been one FDA approved treatment for neurotrophic keratitis (NK): Oxervate (cenegermin-bkbj, Dompe). Before that, there was nothing. Now pharmaceutical options for NK—and persistent epithelial corneal defect (PCED) more generally—in the pipeline number about a half dozen.
Cornea Editor Julie Schallhorn, MD, gave a preview of the articles in the section and talked about the current climate for institutional funding for research.
With dry eye presenting as an increasingly common issue, many companies are exploring options to treat these patients. New products in development are seeking to address different aspects of dry eye, with various amechanisms of action. Three specialists discussed some of these.
Three cornea specialists and one oculoplastics specialist agreed that, in some cases, closing the eyelid with either a permanent or temporary tarsorrhaphy is among the best treatments for corneal healing; the procedure is, in many ways, “the cornea’s best friend.”
“The articles in this issue’s Cornea section focus on challenging situations in cornea and the tools that we have to address them,” said Cornea Editor Julie Schallhorn, MD. She expressed her frustration with the current healthcare environment in meeting the needs of these more challenging patients.
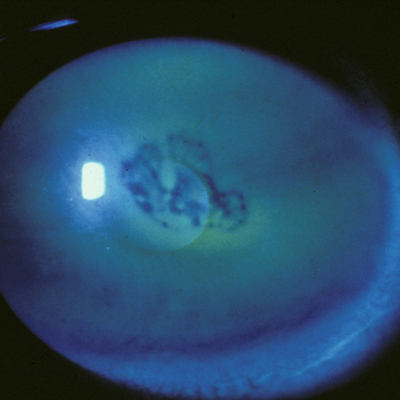
Opacified corneas can present an additional challenge when preparing a patient for cataract surgery. Three physicians discussed how they handle these patients and specific considerations.
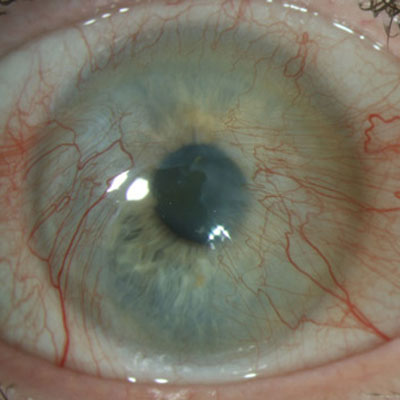
BrightMEM corneal allograft (Brightstar Therapeutics) is a new treatment option for ocular surface disease. Several sources discussed BrightMEM, their experience using it, and its treatment applications.
Many patients use drops to help with a variety of ophthalmic issues, however, one thing to consider is the formulation of these products, specifically if they have preservatives. Three ophthalmologists discussed the precautions with preservatives and shared the importance of monitoring for toxicity and potentially finding alternative treatment options.
In December 2020, the FDA issued “Dry Eye: Developing Drugs for Treatment Guidance for Industry” as a draft for public comment. William Boyd, MD, Deputy Division Director with the FDA’s Center for Drug Evaluation and Research, shared with EyeWorld that the administration hopes to issue its final guidance soon.
Corneal cell therapy is being investigated as a new approach for patients in need of corneal transplantation and treatment for corneal diseases. EyeWorld spoke to several physicians who have experience in this area to hear about specific approaches being developed, current status and results, and the future of these options.