Cornea: Evolving
Winter 2024
by Ellen Stodola
Editorial Co-Director
Currently, corneal cell therapy is being investigated as a new approach for patients in need of corneal transplantation and treatment for corneal diseases. EyeWorld spoke to several physicians who have experience in this area to hear about specific approaches being developed, current status and results, and the future of these options.
Aurion Biotech injectable cell therapy
W. Barry Lee, MD, discussing Aurion Biotech injectable cell therapy, said the exact science behind it is proprietary and unknown. The company takes a donor cadaver cornea and harvests the endothelial cells. The cells are transported to a laboratory where they are expanded in vitro. Once the expansion protocols are completed, the cell may potentially be used for treating up to 100 patients with endothelial dysfunction, Dr. Lee said.
“Cells are delivered to the surgeon on the morning of the surgery and drawn up in a syringe. The patient is prepared in a sterile environment, and the cells are injected into the anterior chamber of the eye after the anterior chamber has been decompressed. A suture is placed in the incision site, and the patient immediately turns over to lie face down for 3 hours. They go home after that time has elapsed with a protective shield over the eye.”
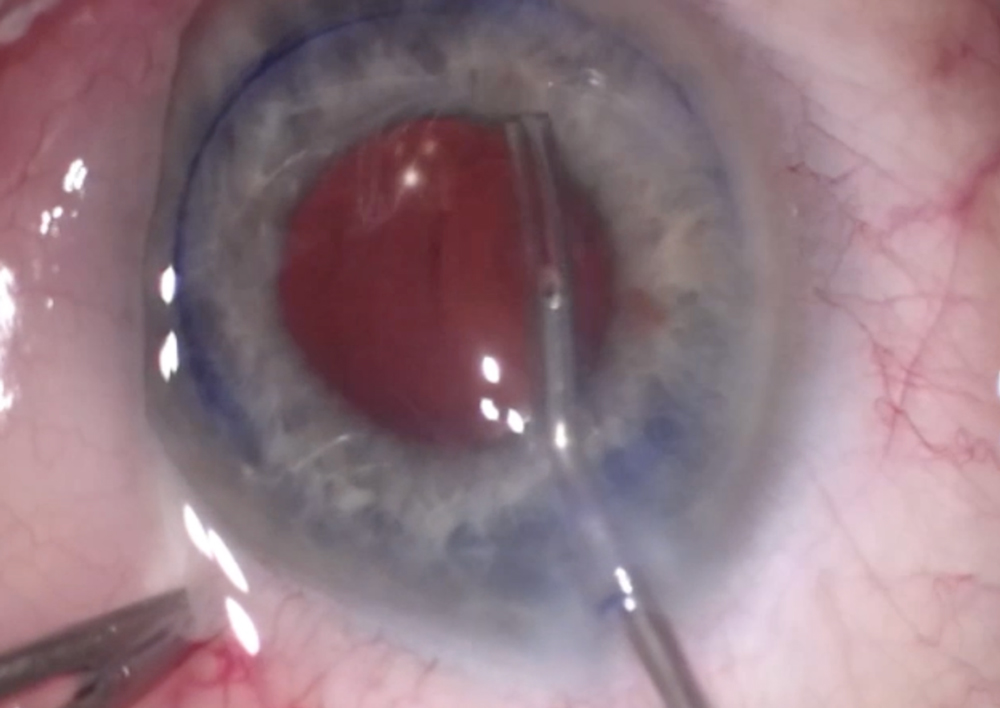
Source: W. Barry Lee, MD
Aurion Biotech has completed their first U.S. clinical trial aimed at treating corneal edema secondary to endothelial dysfunction, Dr. Lee said. The trial combined neltependocel, expanded human allogeneic endothelial cells, with Y-27632, a ROCK inhibitor, in a prospective, multicenter, randomized, double-masked, parallel-arm, cell dose-ranging study. The medications were administered as a one-time intracameral injection in the eye with the primary endpoint representing an improvement of 3 lines of visual acuity at 6 months. While all patients have been treated, the company is awaiting final 6-month results for reporting to the FDA.
Dr. Lee said it’s likely the FDA will review the results of the trial and recommend a Phase 3 clinical trial with the same type of study design. “This trial will likely begin in the second or third quarter of 2025,” he said. “While it is difficult to predict FDA recommendations, it may be 2026 or 2027 before the technique is available or at least approved by the FDA. However, the same technology did receive approval in Japan for clinical use in early 2024.”
The trial for this therapy included patients with endothelial dysfunction with the requirement of pseudophakia and no history of glaucoma, Dr. Lee said. Additionally, he said the majority of patients had Fuchs endothelial corneal dystrophy followed by pseudophakic bullous keratopathy.
“The current standard of care for patients with intractable corneal endothelial cell dysfunction is a cornea transplant, specifically an endothelial keratoplasty (EK),” Dr. Lee said. “The Eye Bank Association of America Statistical Report showed that EK was the most common cornea transplant done in the U.S. in 2023, with 33,715 EK procedures completed, and Fuchs was the most common disease that required a transplant in 2023. If cell therapy works, the need for this number of transplants in the U.S. and across the world would significantly diminish and potentially could replace the need for EK. This could have a major global impact throughout the world.”
Dr. Lee said there are other companies currently developing endothelial cell therapies. He noted that Aurion Biotech is slightly further along in the number of patients treated. Aurion Biotech uses ROCK inhibitors as an addition to the expanded endothelial cells.
“Expanded endothelial cells remain the most exciting new technology for endothelial disease,” Dr. Lee said. “Other devices are in development for countries where tissue is not readily available. These devices are in their infancy but could potentially block edema from entering a diseased cornea, preventing bullous keratopathy and corneal decompensation in countries devoid of tissue for transplantation.”
Emmecell injectable endothelial cell therapy
Ellen Koo, MD, said, with Emmecell, the cells are isolated from donor corneas, and they are expanded in a GMP manufacturing facility in the U.S. Magnetic nanoparticles render the cells magnetic, and they are shipped overnight in syringes to the clinical site. This cell therapy is a simple injection that can be performed at the slit lamp with topical anesthesia and does not require access to an OR, she said. EO2002 cells are injected into the anterior chamber, and a magnetic patch is placed over the eye for a short period of time. Patients are able to resume normal activities within a few hours.
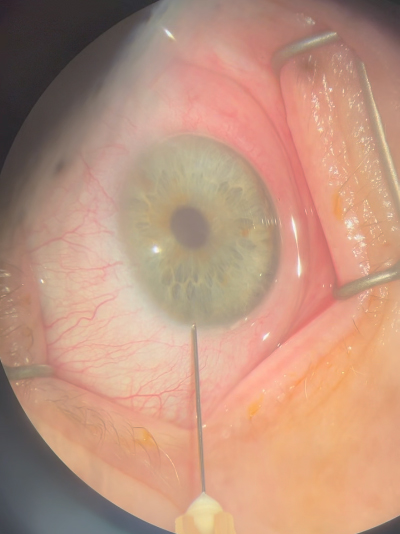
Source: W. Barry Lee, MD
Emmecell has completed enrollment of its randomized controlled clinical trial testing three doses of EO2002, magnetic human corneal endothelial cells. Topline data is expected before the end of this year, and a pivotal trial will launch early next year, Dr. Koo said. She added that there have been no significant safety concerns, and early analysis shows strong signs of efficacy in both BCVA gain and CCT reduction.
With the pivotal trial slated to launch in 2025, Dr. Koo said EO2002 will soon reach many more patients. Availability for use outside of research trials would come after the pivotal trial and FDA approval.
Dr. Koo said the targeted patient population for this therapy are patients with corneal edema due to endothelial dysfunction, secondary to conditions such as Fuchs dystrophy or pseudophakic bullous keratopathy. “These subjects may be candidates for DMEK, DSAEK, DSO, or PKP, but they don’t want the risk associated with these invasive surgeries, or they may not have access to a cornea specialist,” she said. “EO2002 injections will be able to treat patients at an earlier stage of disease, and by a wider provider base of general ophthalmologists or cornea specialists, delaying or even preventing the need for transplant.”
She added that Emmecell’s magnetic cell therapy does not require the surgical step of removing the existing endothelial layer. “This simple injection can be done by any ophthalmologist (not just a cornea specialist), and it can be performed at the slit lamp,” she said. “The recovery is quick with potential for visual gain as early as 1 week. Other treatments require a more invasive surgical procedure, which may result in a slower visual recovery.”
In comparison to current options available for these patients, Dr. Koo said that Emmecell’s magnetic cell therapy is a minimally invasive procedure, requiring no access to an OR, as there is no surgical component. “Patients can resume normal activity the same day and are not required to position for days following the procedure, as is required with current treatments,” she said.
Dr. Koo said she’s excited to see progress in this field by other companies as well, noting the progress of Aurion Biotech’s therapy, which shows that cell therapy will be a game changer. She said that there have been developments beyond magnetic cell therapy for corneal disease, noting that magnetic cell delivery for macular degeneration is in development and stands to restore vision for another large group of patients suffering from vision loss.
Cultivated autologous limbal epithelial cells (CALEC) therapy
Ula Jurkunas, MD, said there are a lot of cell therapy options looking at the corneal endothelium, the inner layer of the cornea. “The cell therapy that we have developed is for corneal epithelium, or the surface cells,” she said. “Corneal epithelial cells get destroyed due to chemical injuries, various infections, genetic disorders, even contact lens wear.” This can destroy the inside, or peripheral corneal epithelial cells, where the corneal stem cells reside.
When there is corneal stem cell destruction, the patient develops limbal stem cell deficiency, the cornea becomes opaque, the patient can become blind, and it can be very painful, Dr. Jurkunas said. “We’ve developed technology where we are harvesting a small biopsy from healthy eyes. Let’s say it’s asymmetric disease—one eye is affected and the other is mostly healthy; we take them to a special GMP lab, and we isolate those stem cells, expand them, and make a lot more of them on a sheet.”
CALEC involves cells on a sheet of amniotic membrane as a scaffold, then they are transplanted to the eye that has the injury or infection. Dr. Jurkunas noted that the first feasibility and safety study was published in Science Advances,1 and the efficacy paper study of the Phase 2 trial has been accepted in Nature Communications. The study showed that 92% of CALEC grafts achieved partial or complete success at 18 months of follow-up.
The trial was the first of this kind in the U.S., though cultivated epithelial stem cells have been used outside the U.S. in the past. “We were the first center to develop this trial, and we did the trial without the support of industry,” she said. She added that it was the first trial of this kind using epithelial cells that are autologous, so there isn’t any rejection.
Dr. Jurkunas said that next step will be to go to the FDA to determine if a Phase 3 clinical trial is needed. Most therapies require a Phase 3 trial, she said, adding that the therapy has so far shown that it’s safe and feasible.
One key factor still to be determined is where manufacturing for this therapy would occur—at one center or multiple centers in the country. It does require quite a lot of resources to manufacture it, she said.
Dr. Jurkunas noted that this therapy is different from a standard corneal transplant. With the disease, a standard corneal transplant is going to fail right away because you don’t have healthy stem cells, so patients in need of this therapy may need a regular transplant and this transplant together.
Looking to future developments with this treatment, Dr. Jurkunas said a next step could be investigating how to do this with allogeneic cells, for patients who have bilateral LSCD. A treatment like this would take cells from a cadaveric donor and culture, possibly with similar methods or with modifications. This would be helpful, she said, because of the prevalence of bilateral cases, but by using another donor, you would need to factor in the possibility of rejection as well.
This is a stepping stone for cell therapy in the U.S., she said. “Our regulatory requirements are stringent, and this is the first time we’re using stem cell therapy in cornea.”
She’s interested to see the other therapy options being developed for endothelial cells as well. If you have a cell therapy for endothelial cells, you wouldn’t need as many corneal cells or cadaveric donors.
“There are regulatory hurdles that one has to overcome, but once we have one product, it’s always easier for other products to get approved and other technologies to be developed, so I think it’s definitely a stepping stone for that,” she said.
ARTICLE SIDEBAR
Albert Jun, MD, PhD, also commented on the future of this field, noting that it’s an exciting time for cornea and cell therapy. “I think part of what makes it so interesting is the very focused type of pathology that we sometimes see in the cornea. The accessibility of tissue also makes it appealing. It’s the extension of that minimally invasive approach for everything that we do in medicine to make things better for patients, to do less but accomplish the goal that the patient needs to have restored function.”
Dr. Jun called the developments in corneal cell therapy a logical extension to the lamellar keratoplasty trend. Using technology to produce and grow cells from a source, whether it’s a patient’s own cells or a donor, helps with the targeted ability to treat the patient’s vision loss, he said, adding that he began doing work in this area around 10 years ago.
There’s a general concept of putting endothelial cells back onto the cornea where they need to be to work, he said, adding that tremendous work has been done in this area by Shigeru Kinoshita, MD, PhD. “Other technologies have been developed around cell injection, taking the cells you grow, injecting them into the eye, and having them stick to the cornea and work,” Dr. Jun said.
The next question to look at is how well this technology works compared to current methods of endothelial keratoplasty. “I think cell injection proved a lot of things,” he said. “It proved you can take cells off donor corneas and grow them, they retain their function, you can process them safely and put them back into a person, and they can work. That’s been proven, and that’s tremendous. But what the field will determine is whether or not that approach is advantageous over the current way of doing things.” Dr. Jun said it will also be important to see how this treatment works in a variety of conditions.
The other issue is efficacy. “We have to see if the results are just as good as or better than DMEK, and that’s something that patient experience will teach us.” Dr. Jun added that cost will be a factor as well.
Dr. Jun has worked with the eye bank VisionGift in Portland, Oregon, and OcuCell to develop these cell technologies. OcuCell’s Endo-Tek is human-derived cultured corneal endothelial cells seeded onto a biocompatible hydrogel. “Our approach has been to replace the membrane and the cell with a synthetic membrane that’s a replacement for Descemet’s membrane, and this has cells on it,” he said.
About the physicians
Albert Jun, MD, PhD
Vernah Scott Moyston Professor and Chair
Department of Ophthalmology
University of Virginia School of Medicine
Charlottesville, Virginia
Ula Jurkunas, MD
Professor of Ophthalmology
Harvard Medical School
Boston, Massachusetts
Ellen Koo, MD
Associate Professor of Ophthalmology
Bascom Palmer Eye Institute
University of Miami
Health System
Miami, Florida
W. Barry Lee, MD, FACS
Cornea Fellowship Director
Eye Consultants of Atlanta
Atlanta, Georgia
Reference
- Jurkunas UV, et al. Cultivated autologous limbal epithelial cell (CALEC) transplantation: development of manufacturing process and clinical evaluation of feasibility and safety. Sci Adv. 2023;9.
Relevant disclosures
Jun: OcuCell
Jurkunas: Ocucell
Koo: Emmecell, KOWA
Lee: Aurion Biotech
Contact
Jun: CFJ3PJ@uvahealth.org
Jurkunas: Ula_Jurkunas@meei.harvard.edu
Koo: exk126@med.miami.edu
Lee: wblee@mac.com




