All articles from EyeWorld.org on the topic of cataract surgery. EyeWorld is the award-winning member publication of ASCRS. The magazine provides news and updates from the Society as well as clinical features in the areas of cataract, refractive, cornea, glaucoma, and practice management. It publishes quarterly with editorial direction from its medical editorial board and staff.
➤ Phase 3 trial investigating inflammation, pain management after cataract surgery
➤ Positive topline data in study for RP gene therapy
➤ Study: Intravitreal cancer drug found to preserve vision more than traditional therapy
➤ Several studies show efficacy of home-based OCT
➤ Company announces expense reduction and refocus plan
➤ ASCRS news and events
➤ Cholesterol-lowering gene linked to risk of cataract development
➤ Study: Structure, size of individual’s visual cortex affects visual potential
➤ Purkinje reflections for capsulotomy centration
➤ Possible new therapeutic approach to treat retinitis pigmentosa
➤ ASCRS news and events
➤ FDA 510(k) clearance granted to FLACS system
➤ New genetic eye disease discovered
➤ Primary endpoint met in Phase 3 trial for dry eye drug candidate
➤ New analysis of thyroid eye disease therapy
➤ Licensing agreement for commercialization of investigational myopia therapeutic
➤ ASCRS news and events
➤ FDA approves brolucizumab injection for DME
➤ NDA filed for geographic atrophy drug
➤ Study: Dietary supplements can slow AMD progression
➤ First global study evaluates AI for diabetic retinopathy and referral uptake
➤ Plans to begin two Phase 3 trials for investigational thyroid eye disease therapy
➤ ASCRS news and events
➤ Phase 4 results evaluating intravitreal implant
➤ Interim results for X-linked retinitis pigmentosa treatment
➤ New non-contact glaucoma laser treatment receives CE mark
➤ Companies collaborate for regenerative dry eye therapy
➤ Complimentary MIGS video series
➤ ASCRS news and events
Two optometrists discussed their experiences in practices that have a comanagement approach. They shared recommendations for practices interested in expanding their comanagement network.
➤ FDA approves image and data management/analysis platform
➤ Results from pivotal Phase 3 trial for new Demodex treatment
➤ Trial demonstrates safety, efficacy of mydriasis-reversal drug in pediatric patients
➤ Phase 3 pivotal trial for novel dry eye disease, MGD drug candidate
➤ Phase 3 results for investigational geographic atrophy therapy
➤ New coalition supports sustainability in ophthalmology
➤ ASCRS news and events
➤ Phase 3 clinical trials complete for ‘superpotent’ corticosteroid
➤ New rapid, quantitative MMP-9 tear test
➤ First pediatric patient dosed with experimental gene-editing therapy for Leber congenital amaurosis
➤ Study: Comparison of NCX 470 to latanoprost for glaucoma
➤ Collaborations for inherited retinal diseases
➤ ASCRS news and events
➤ Topline data from Phase 3 trial of presbyopia-correcting drop
➤ FDA gives 510(K) approval to updated neuromodulation therapy for increased tear production
➤ Enrollment complete for Phase 3 trial of reproxalap
➤ DME trial to focus on underrepresented patient populations
➤ BLA submitted to FDA for wet AMD therapy
➤ Companies collaborate to further develop gene therapy for retinal disease
➤ ASCRS news and events
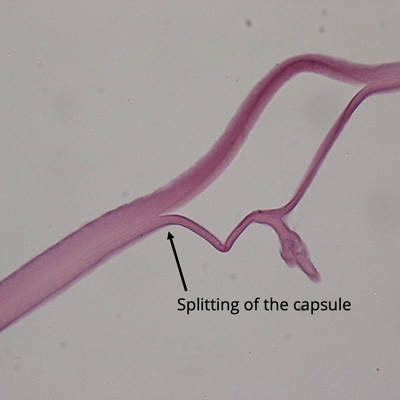
Dead bag syndrome, first described by Samuel Masket, MD, is the subject of a recent paper published in the Journal of Cataract & Refractive Surgery (JCRS). The study’s corresponding author, Liliana Werner, MD, PhD, described the paper as the first formal peer-reviewed study on dead bag syndrome. It examined the clinical and histopathological findings of 10 suspected cases.