Browse articles from EyeWorld.org related to glaucoma. EyeWorld is the award-winning member publication of ASCRS. The magazine provides news and updates from the Society as well as clinical features in the areas of cataract, refractive, cornea, glaucoma, and practice management. It publishes quarterly with editorial direction from its medical editorial board and staff.
MIGS procedures have become a mainstay in the treatment of mild to moderate glaucoma, but what pressures can physicians realistically expect with these? Three surgeons spoke about this, as well as how MIGS can fit into the overall glaucoma management process and what to do if MIGS procedures don’t achieve the desired IOP.
There is a significant focus on stopping progression of optic nerve damage and subsequent vision loss in glaucoma, but what about patients who have already reached a point of vision loss? What modern visual rehabilitation technologies and services are available to them? EyeWorld spoke with three experts on this topic.
➤ First in-human, non-invasive femtosecond laser trabeculotomy data
➤ Special Protocol Assessment received for Phase 2b/3 trial for dry AMD treatment
➤ FDA gives clearance to IND for geographic atrophy treatment
➤ Positive preliminary data in trial for retinitis pigmentosa and Leber congenital amaurosis therapy
➤ Wet AMD treatment proceeds to Phase 2 trial
➤ ASCRS news and events
Patients with glaucoma who need cataract surgery have many lens options available, and it’s important for physicians to present the potential benefits and risks so patients can make the best decision. Two experienced ophthalmologists discussed different lens options for glaucoma patients and important factors to consider.
ASCRS Subspecialty Day will feature full-day programming in the areas of cornea, glaucoma, and refractive surgery. New this year, registrants of Subspecialty Day will also be able to attend the inaugural Digital Day program, taking place in conjunction with ASCRS Subspecialty Day and sponsored by the Digital Ophthalmic Society.
Glaucoma Editor Manjool Shah, MD, talks about what readers can expect in the Glaucoma section of the issue, as well as the value of ASCRS and the Annual Meeting. He also welcomes the newest members of the Glaucoma Editorial Board.
➤ Results from investigational non-incisional glaucoma treatment
➤ Study: preservative-free latanoprost efficacy similar to preserved alternative
➤ FDA accepts priority review NDA for vitreoretinal lymphoma treatment
➤ Companies partner to develop dry eye drop
➤ Company announces lead asset in its agonist antibody pipeline
➤ ASCRS news and events

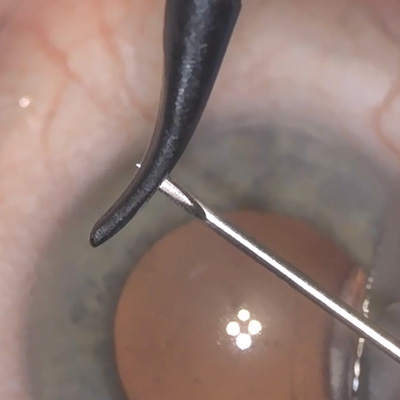
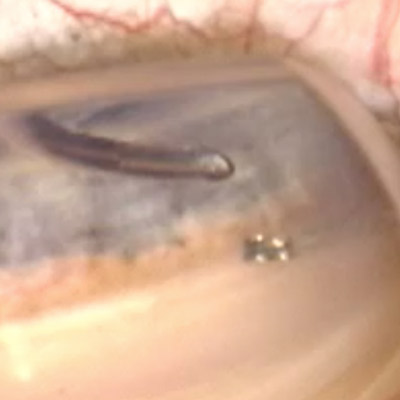
In the trial, 12-month mean IOP was lowered 40% from baseline with the biostent, with 80% of patients achieving at least a 20% reduction in IOP.
Source: Sean Ianchulev, MD, MPH
The first-in-human trial and data for suprachoroidal outflow enhancement using a novel micro-interventional technology and first-in-class biostenting approach developed by Sean Ianchulev, MD, MPH, was published online in the British Journal of Ophthalmology in January.
“A fundamental and unanswered question concerning all MIGS devices is whether they have any measurable ability to help POAG patients retain their vision.” The authors of a study published in the American Journal of Ophthalmology sought to answer this question. The study is a post-hoc visual field analysis of the 5-year HORIZON trial.
➤ NDA submitted for sustained-release travoprost implant
➤ BLA for wet AMD, DME, DR treatment accepted with priority review
➤ Topline results from 12-month safety clinical trial for investigational dry eye drug
➤ Companies collaborate for research on sustained release for investigational GA drug
➤ ASCRS news and events