Browse articles from EyeWorld.org related to presbyopia. EyeWorld is the award-winning member publication of ASCRS. The magazine provides news and updates from the Society as well as clinical features in the areas of cataract, refractive, cornea, glaucoma, and practice management. It publishes quarterly with editorial direction from its medical editorial board and staff.
➤ FDA approves new ocular surface anesthesia gel
➤ New drop approved to reduce IOP
➤ Newly available IOL incorporates technology that minimizes visual disturbances
➤ Interim data from Phase 1 trial for intravitreal implant to treat AMD
➤ Multiparty financing solution brought to ophthalmic practices
➤ Complimentary access to video journal
➤ ASCRS news and events
➤ Study: Specific form of AMD linked to high risk of heart disease
➤ Primary endpoints achieved in study of dry eye therapy
➤ First patient enrolled in study for new pharmacologic presbyopia therapy
➤ Company submits IND for advanced AMD treatment candidate
➤ Acquisition of topical dosing technology
➤ Patent granted for oral drug candidate targeting retinal diseases
➤ Complimentary access to video journal
➤ ASCRS news and events
Refractive Editor Vance Thompson, MD, expresses his enthusiasm about the recent ASCRS Annual Meeting and previews the articles in the section.
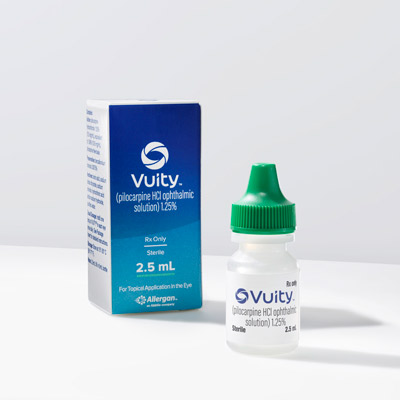
Drops that temporarily relieve the symptoms of presbyopia are an exciting new technology, with the first product approved in this space late last year. Two physicians shared their early experience with Vuity (pilocarpine, Allergan).
“X-Rounds: Refractive Cataract Surgery to the Max” is a fast-paced session at the ASCRS Annual Meeting where members of the panel presented their answer to a question or thoughts on a topic in 2.5 minutes or less. The audience then voted for their favorite in each section.
➤ Topline data from Phase 3 trial of presbyopia-correcting drop
➤ FDA gives 510(K) approval to updated neuromodulation therapy for increased tear production
➤ Enrollment complete for Phase 3 trial of reproxalap
➤ DME trial to focus on underrepresented patient populations
➤ BLA submitted to FDA for wet AMD therapy
➤ Companies collaborate to further develop gene therapy for retinal disease
➤ ASCRS news and events
➤ Launch of Phase 3 clinical trial for presbyopia drop candidate
➤ Topline results for investigational dry eye therapy
➤ Positive findings for photobiomodulation dry AMD treatment
➤ Enrollment completed for investigational oral diabetic retinopathy therapy
➤ Longer-term data from Phase 3 studies evaluating geographic atrophy therapy released
➤ Orphan drug designation granted to investigational retinitis pigmentosa treatment
➤ ASCRS news and events
➤ Results from pivotal amblyopia trial
➤ Phase 2 clinical program for third-generation keratoconus treatment
➤ Study compares tolerability of standard drop vs. novel drug delivery device
➤ Phase 3 clinical studies for investigational presbyopia drop complete
➤ Orphan drug designation granted for fungal keratitis treatment
➤ Full rights regained for investigational dry AMD therapy
➤ ASCRS news and events
➤ Launch of IOL made with new ‘glistening-free’ material
➤ Enrollment complete in study of mydriasis-reversing drop in a pediatric group
➤ Acquisition of electrophysiology medical device company
➤ Licensing of investigational neuroprotective drug
➤ ASCRS news and events
➤ FDA approves first bispecific antibody for the eye
➤ FDA approves treatment for rare uveal melanoma
➤ New data on clinical programs investigating phentolamine ophthalmic solution
➤ Phase 2 clinical trial results published for investigational drug targeting allergic conjunctivitis
➤ Patient dosing begins for gene therapy to treat X-linked retinitis pigmentosa
➤ New effort to research glaucoma
➤ ASCRS news and events