Browse articles on EyeWorld.org. EyeWorld is the award-winning member publication of ASCRS. The magazine provides news and updates from the Society as well as clinical features in the areas of cataract, refractive, cornea, glaucoma, and practice management. It publishes quarterly with editorial direction from its medical editorial board and staff.
While ophthalmologists agree that glaucoma drops are not likely to go extinct as part of glaucoma management, recent innovation continues to shift their place in many physicians’ glaucoma treatment algorithm. One of these disruptive innovations is sustained drug delivery.
Two experts weighed in on the topic.
ASCRS Annual Meeting Program Chair Thomas Samuelson, MD, spoke to EyeWorld about the upcoming Annual Meeting and what he’s looking forward to in Los Angeles, California.
EyeWorld honors the lives of ophthalmologists who died in 2024 with recognition here.
In this issue’s column, the authors discussed the surgical work of ophthalmologists at Aravind Eye Hospital in India. They shared the results of the standardized protocols that include reusing devices for cataract surgery usually deemed single use in Western countries.
I. Howard Fine, MD, a past president of ASCRS and innovator of surgical instruments and procedures, died August 29, 2024, at 88 years old. Several physicians paid tribute to and shared their memories of Dr. Fine.
While true allergies to materials used during cataract surgery are uncommon, this is a topic that physicians may need to discuss with patients. Two physicians highlighted some of the key concerns they hear from patients and how they approach these to identify real allergies versus intolerances.
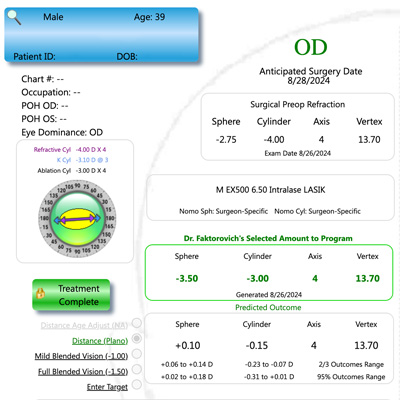
What’s the latest tool in the refractive surgeon’s toolbox? As with many fields from marketing to medicine, it’s artificial intelligence (AI). Several surgeons discussed how AI is impacting refractive surgery, as well as some other areas of the anterior segment.
Many patients use drops to help with a variety of ophthalmic issues, however, one thing to consider is the formulation of these products, specifically if they have preservatives. Three ophthalmologists discussed the precautions with preservatives and shared the importance of monitoring for toxicity and potentially finding alternative treatment options.
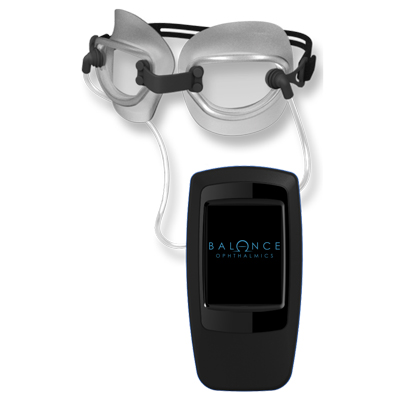
In June 2024, the FDA approved the FSYX Ocular Pressure Adjusting Pump by Balance Ophthalmics, making it the first non-surgical, non-pharmaceutical, IOP-lowering therapy for patients with open angle glaucoma. Physicians shared how they are excited about being able to lower IOP safely in normal tension glaucoma patients with accompanying nocturnal IOP elevations.
In December 2020, the FDA issued “Dry Eye: Developing Drugs for Treatment Guidance for Industry” as a draft for public comment. William Boyd, MD, Deputy Division Director with the FDA’s Center for Drug Evaluation and Research, shared with EyeWorld that the administration hopes to issue its final guidance soon.