Refractive
Winter 2024
by Liz Hillman
Editorial Co-Director
What’s the latest tool in the refractive surgeon’s toolbox? As with many fields from marketing to medicine, it’s artificial intelligence (AI). EyeWorld spoke with several surgeons to learn more about how AI is impacting refractive surgery, as well as some other areas of the anterior segment.
“The field of refractive surgery has become incredibly complex,” said Ella Faktorovich, MD. There are different types of refractive procedures and important diagnostic tools coinciding with preop screening. “How do we decide, for example, whether a patient with mild inferior corneal steepening is best treated with a corneal or lens procedure? Is the steepening consistent with keratoconus and significant enough to benefit from crosslinking prior to refractive surgery? When planning the patient’s treatment, how can we use postoperative outcomes data from other patients who underwent similar treatments to plan this patient’s treatment? AI has become essential to help us answer these questions and navigate the increasingly complex field of refractive surgery diagnostics and treatments.”
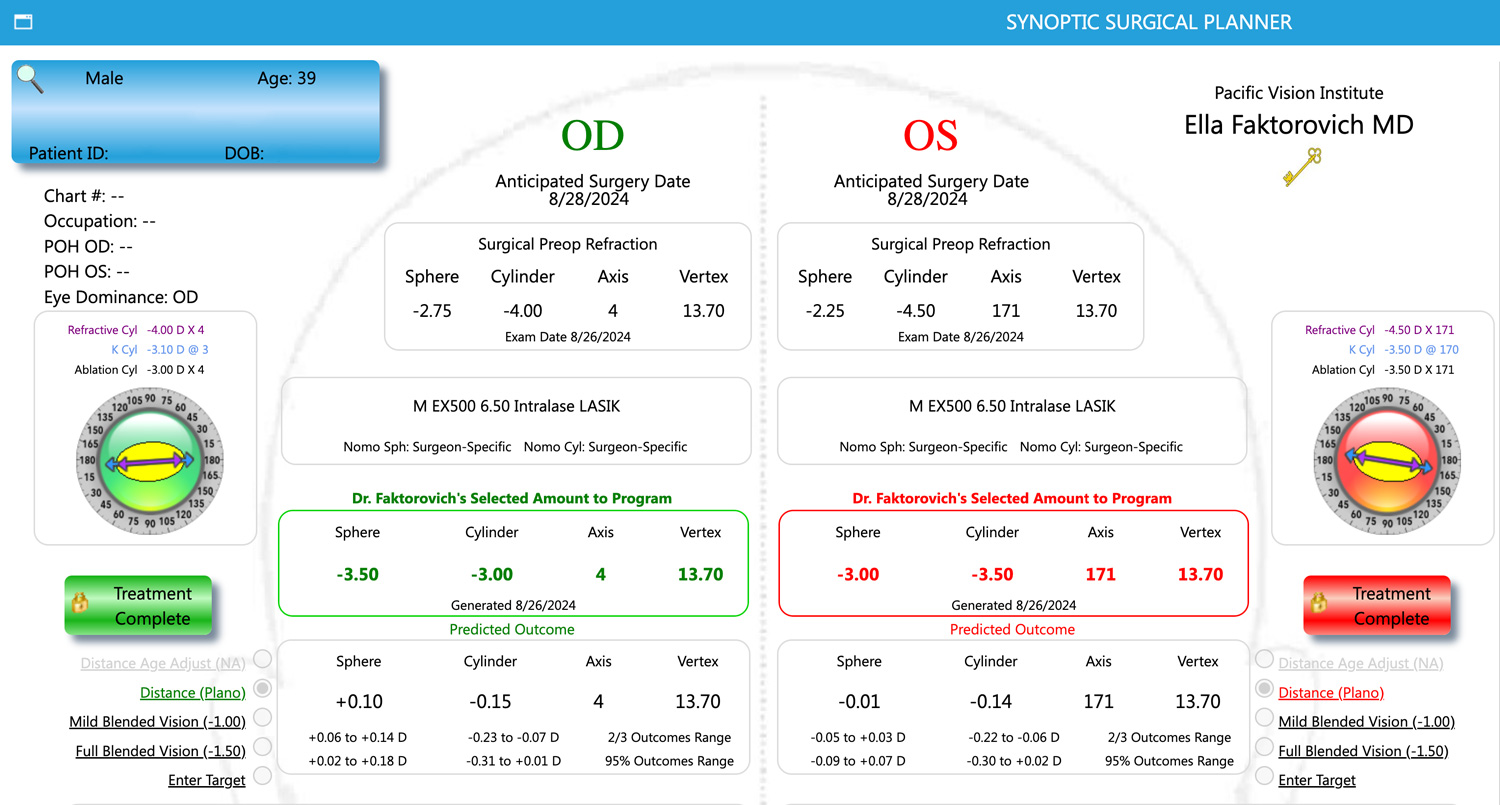
Source: Ella Faktorovich, MD
AI in diagnostics
“While the journey toward widespread use is in its early stages, the path forward is clear and filled with promise, emphasizing the importance of ongoing research, education, and technological development in refractive surgery.”
Renato Ambrósio Jr., MD, PhD
To determine a patient’s suitability for refractive surgery, various ocular parameters need to be carefully measured and analyzed, Dr. Faktorovich said. “We typically use five different methods to assess corneal health—topography, tomography, epithelial thickness mapping with widefield OCT, corneal biomechanics, and aberrometry to map higher order aberrations. Diagnostic software in topography and tomography uses [rule-based] AI to benchmark each individual’s corneal characteristics against the database in the software. Tomography performed with Pentacam [Oculus], for example, generates seven different corneal indices. The device’s AI then compares these indices to the software’s database and determines the likelihood of keratoconus. If keratoconus is likely, the software grades the severity. Currently, only topography and tomography devices have AI capabilities.
“Devices such as the Ocular Response Analyzer [Reichert] to measure corneal biomechanics and OCT to perform epithelial thickness mapping don’t have AI,” she continued. “A clinician relies on their expertise and published data to determine whether the results of these tests are normal or abnormal. This is an area where technology can be improved to include AI. Additionally, there could be improvement in creating a software that integrates information from multiple testing devices and benchmarks that against a data set of normal and abnormal corneal tests performed with these multiple devices. This multimodal benchmarking could be especially helpful when findings from one or several devices are mildly abnormal.”
AI for ectasia risk screening
Renato Ambrósio Jr., MD, PhD, said Stephen Klyce, PhD, led groundbreaking work in the 1990s that used AI to help diagnose keratoconus.1 Since then, AI in refractive surgery screening has expanded.
“The characterization of the inherent susceptibility of the cornea for biomechanical decompensation and ectasia progression2-4 has to be considered along with the impact of the LVC procedure.5 While this concept relates to the two-hit hypothesis, including intrinsic and extrinsic factors,6 AI’s ability to analyze complex datasets and identify subtle patterns by considering multiple features instantaneously makes it a valuable tool for improving accuracy and inclusivity in refractive procedures. This individualized approach improves sensitivity for safety and specificity for higher inclusivity of refractive procedures,” Dr. Ambrósio said.
Dr. Ambrosio said we don’t have data about how widespread AI use is for ectasia risk assessments in refractive surgery screening, but he thinks it’s limited. “Ectasia is a very severe and feared complication of LVC. While detailed data on its prevalence is not fully available, its incidence has reduced. The potential for AI to enhance ectasia risk assessment is significant,” he said, adding that adopting AI-based ectasia risk assessment tools is an opportunity to improve clinical practice. “Key factors include increasing awareness and education among clinicians, obtaining regulatory approval, ensuring data privacy and security, and developing cost-effective solutions. Addressing these barriers can promote the benefits of AI, leading to broader adoption and enhanced patient safety.”
Though rare, Dr. Ambrósio said he thinks AI for determining ectasia risk scores is an “absolute must-have in refractive surgery.” Besides LVC, keratoconus detection is essential in refractive cataract surgery as it impacts IOL selection, quality of vision, and the strategy for corneal enhancements.
While Dr. Ambrósio noted the utility of AI in refractive surgery screening overall, he also provided some more specific updates.
“The TBIv2 (or BrAIN-TBI) was optimized based on a larger dataset for training and including novel parameters.7 Further optimization is expected from integrating segmental tomography data with Fourier-domain OCT, with epithelial and Bowman thickness profiles,” he said. “Genetic testing and other molecular biology tests may also play a relevant role.
“Considering the impact of surgery, we developed the RTA (relational thickness altered) with an AI algorithm that includes the patient’s age, thinnest point data, ablation depth (PRK or LASIK), flap (LASIK), or cap thickness and lenticule extraction (LALEX/SMILE). The RTA considers each case individually and weighs the flap or cap and the ablation in LASIK differently. It provides a superior approach to characterizing the lamellar dissection and ablation, significantly outperforming traditional methods of calculating residual stromal bed and percentage of thickness altered for a more comprehensive and accurate risk assessment.”
The updates and developments in AI for refractive surgery are driven by a desire for increased patient safety, Dr. Ambrósio said. “By prioritizing safety and leveraging advanced AI algorithms like the RTA and the TBI, we can generate the enhanced susceptibility score available in the BrAIN Enhanced Corneal Ectasia Software,” he said. “This individual approach combines a nuanced understanding of the surgical impact and the intrinsic susceptibility.
“Nevertheless, preventing ectasia after refractive surgery (and in non-refractive patients) should include educating patients on not rubbing the eye. When ancient and artificial intelligence is consciously applied, we can uphold the highest standards of patient care and significantly enhance refractive surgery,” Dr. Ambrósio said.
Nambi Nallasamy, MD, who is more involved in research for AI applications in cornea (see the box on page 56), said we can always improve upon screening for refractive surgery, and AI is a promising avenue.
“I’m sure that AI will be able to identify patients better than we can alone. It may require other types of imaging,” he said. “I think the combination of more data, more types of imaging plus AI is really going to help us here.”
Travis Redd, MD, also said that the primary application for AI in refractive surgery at the moment is in screening for ectasia risk.
“Deep learning models are very good at detecting subtle patterns in images such as corneal tomography,” he said. “This capability could be used to preoperatively identify patients with a high likelihood of developing post-refractive surgery ectasia.”
AI in treatment planning
The nomograms used for refractive surgery planning are heavily dependent on machine learning, which takes outcomes from cases and updates the formulas, Dr. Faktorovich said.
“To fully harness the power of treating both lower and higher order aberrations, nomogram use is essential. AI is, therefore, a critical component of achieving the best vision postoperatively,” she said. “We use both [machine learning] AI and [rule-based] AI to plan topography-guided surgical treatments. With [machine learning] AI, linear regression analysis formulas allow us to accurately plan treatment of sphere, cylinder, and higher order aberrations such as spherical aberration, coma, trefoil and others. With [rule-based] AI, we can integrate data from the corneal aberrometer and topographer to help not only decide if the patient would benefit from topography-guided treatment but also what is the correct treatment to program into the laser to achieve the most precise outcomes. Postoperative outcomes data on visual acuity, refractive error, and higher order aberrations are then input into another AI-driven software to generate nomograms for subsequent patients undergoing treatments.”
In the future, Dr. Faktorovich said she envisions AI being used for enhanced diagnostics. Dr. Redd said there are a few things that are needed to bring AI in refractive surgery (and the anterior segment, in general) to the next level.
“Deep learning models have been developed to successfully perform image-based diagnosis of several anterior segment diseases, including keratoconus, infectious keratitis, pterygia, and trachoma among many others. AI models have also been trained to provide quantification of various biomarkers of disease severity, allowing more objective monitoring of progression and response to treatment over time,” he said. “However, currently the only FDA-authorized AI-enabled SaMD (software as a medical device) in ophthalmology are for automated diagnosis of diabetic retinopathy. There are many reasons for the gap between the large number of AI models described in the ophthalmology literature and the few that have been implemented clinically, but chief among them are the scarcity of representative datasets for training and evaluating AI models, limited interdisciplinary collaboration, and lack of well-defined reimbursement models.”
Article Sidebar
Other AI updates
Dr. Nallasamy shared some insights on where AI is being applied to other areas of the anterior segment. He said there is currently a clinical trial being led by Maria Woodward, MD, looking at microbial keratitis. The work is developing algorithms to identify key parameters involved in corneal ulcers. This research, he said, could help provide some objectivity in a world that’s usually subjective.
“Typically, we’ve done slit lamp exams on these patients. Often we didn’t even take photos, relying on just seeing whether it got better or worse and changing treatment accordingly, but with these tools, we’ll be able to rigorously follow the size and response to treatment and titrate accordingly,” he said.
Dr. Nallasamy said his lab is doing work using confocal microscopy and AI to develop a system that can automatically diagnose the type of corneal infection earlier on, rather than just tracking its response to treatment.
Separately, Dr. Nallasamy said his lab is also creating an intraoperative decision-making tool for cataract surgery that, using AI, will help the surgeon better understand the likelihood of a patient potentially needing a pupil expansion device during their procedure, based on how the eye is responding in real time to the surgery.
Also in the anterior segment, Dr. Nallasamy referenced work being done by Carol Karp, MD, using anterior segment OCT and AI to develop a system that can differentiate OSSN from benign tumors.
About the physicians
Renato Ambrósio Jr., MD, PhD
Adjunct Professor of Ophthalmology
Federal University of the State of Rio de Janeiro
Rio de Janeiro, Brazil
Ella Faktorovich, MD
Pacific Vision Institute
San Francisco, California
Nambi Nallasamy, MD
Assistant Professor of Ophthalmology and Visual Sciences
Assistant Professor of Computational Medicine and Bioinformatics
University of Michigan
Kellogg Eye Center
Ann Arbor, Michigan
Travis Redd, MD
Assistant Professor of Ophthalmology
School of Medicine
Oregon Health & Science University
Portland, Oregon
References
- Maeda N, et al. Neural network classification of corneal topography. Preliminary demonstration. Invest Ophthalmol Vis Sci. 1995;36:1327–1335.
- Ambrosio Jr R, Randleman JB. Screening for ectasia risk: what are we screening for and how should we screen for it? J Refract Surg. 2013;29:230–232.
- Ambrósio Jr R, Belin M. Enhanced screening for ectasia risk prior to laser vision correction. Int J Keratoconus Ectatic Corneal Dis. 2017;6:23–33.
- Ambrósio Jr R. Post-LASIK ectasia: twenty years of a conundrum. Semin Ophthalmol. 2019;34:66–68.
- Dupps WJ, Seven I. A large-scale computational analysis of corneal structural response and ectasia risk in myopic laser refractive surgery. Trans Am Ophthalmol Soc. 2016;114:T1.
- McGhee CNJ, et al. Contemporary treatment paradigms in keratoconus. Cornea. 2015;34 Suppl 10:S16–23.
- Ambrósio Jr R, et al. Optimized artificial intelligence for enhanced ectasia detection using Scheimpflug-based corneal tomography and biomechanical data. Am J Ophthalmol. 2023;251:126–142.
Relevant disclosures
Ambrósio: Alcon, Ambrósio Vision Academy, Brazilian Artificial Intelligence Networking in Medicine (BrAIN), Carl Zeiss Meditec, Mediphacos, Oculus
Faktorovich: None
Nallasamy: Recordati Rare Diseases
Redd: None
Contact
Ambrósio: dr.renatoambrosio@gmail.com
Faktorovich: ella@pacificvision.org
Nallasamy: nnallasa@med.umich.edu
Redd: redd@ohsu.edu




