Glaucoma Bonus Issue
February 2025
by Liz Hillman
Editorial Co-Director
While ophthalmologists agree that glaucoma drops are not likely to go extinct as part of glaucoma management, recent innovation continues to shift their place in many physicians’ glaucoma treatment algorithm. One of these disruptive innovations is sustained drug delivery.
There are currently two sustained-release drugs for IOP lowering on the market in the U.S., as well as several in the pipeline.
Blake Williamson, MD, said the currently available and pipeline of sustained-release glaucoma therapies “highlight the number one problem that we face as clinicians [with glaucoma] … which is compliance.”
“With our patient population, a lot of times they can be older, have a lot of comorbidities, and a lot of other things going on in their life, and taking daily medications and eye drops can be very laborious. It can be dangerous in some cases. Sometimes I’ll see patients who are taking their steroid drop instead of taking their glaucoma drop. Or patients are poking themselves in the eye, contaminating the dropper. All of those things I see routinely in my patient population,” Dr. Williamson said. “At this point, I think the science and the industry side have collaborated to figure out how can we take this out of our patients’ hands and get them drugs safely and efficaciously. I think that’s why you’re seeing drug delivery innovation across all different spectrums and all different anatomical parts of the eye. Retina has been leading the way with that for years and it’s been fantastic, and I think we’re catching up on the anterior segment side.”

Source: Blake Williamson, MD
Current experience
Dr. Williamson said he has experience with both commercially available sustained-release glaucoma implants: Durysta (bimatoprost intracameral implant, AbbVie) and iDose TR (travoprost intracameral implant, Glaukos).
Dr. Williamson said when it comes to patient selection, in some cases it’s easy. If the patient has already had a MIGS procedure that stripped the trabecular meshwork, surgeons may not be comfortable anchoring iDose TR, making Durysta a more suitable option. He said Durysta may also be more suited for phakic patients. “For me, that’s a place where Durysta might make a little bit more sense, being not as invasive, not as big inside the eye,” he said.
If the patient is having cataract surgery, Dr. Williamson said iDose TR makes sense, especially because it seems to have a longer duration per the clinical trials. “Maybe there are nursing home patients where you’re concerned with their ability to get to appointments, their ability to follow up, you’re not sure when you’re going to see them again, and you may want to see them with something longer lasting,” he said.
In all of his examples, Dr. Williamson said either Durysta or iDose TR would be a fine option, but one or the other may make more sense, depending on your personal algorithm.
Jacob Brubaker, MD, said his preferred algorithm overall is SLT followed by the addition of a topical prostaglandin, if needed, to see how it works for the patient. “If the patient has difficulty remembering or tolerating, I will have a low threshold for moving on to iDose TR,” he said. “It’s designed to last 2–3 years, and while we don’t have FDA approval to replace them yet, hopefully in the next 2–3 years we gain that ability to redose. I think that’s what Durysta was missing; it was only able to be dosed once on label. … I do think the longevity and the safety of iDose TR is a game changer.”
A 2023 randomized, double-masked, multicenter, Phase 2 trial evaluating the long-term safety and efficacy of iDose found that the implant was able to maintain IOP lowering and reduce IOP-lowering medication burden for up to 36 months.1
Laboratory studies of Durysta showed that drug release from the implant was completed by 90 days, and “pharmacokinetics studies” in an animal model showed tissue drug levels were below limits of detection by 4.2 months.2 The efficacy of the treatment may be longer, however. Teymoorian et al. found in a retrospective single-site study that the “mean IOP was 16.6 mm Hg at baseline and 13.3 mm Hg at 11–13 months, with the mean number of topical IOP-lowering medications used reduced from 1.4 at baseline to 0.2 at 11–13 months,” with a single administration of the bimatoprost implant.3
Dr. Williamson doesn’t yet do a combined MIGS/sustained-release implant procedure but rather waits to see how the patient does after the MIGS. “Let’s say I’m going to place some iStent infinites [Glaukos]; we can see how they do, maybe they went from three drops to two drops or two drops to one drop, and I’m just wanting to get those last few IOP points to get them to a dropless situation, maybe then I would go back and place the iDose TR,” he said.
In the pipeline

Source: Jacob Brubaker, MD
There are several companies still developing sustained-release IOP-lowering therapies at various stages (this is not an exhaustive list):
- PAXTRAVA (OTX-TIC, Ocular Therapeutix) is an implant of “travoprost-loaded microparticles embedded in ELUTYX,” a “bioresorbable, programmable hydrogel matrix that encapsulates drug to provide sustained and localized delivery,” according to the company’s website. Positive data from its Phase 2 clinical trial was reported in April 2024.
- LL-BMT1, a preservative-free, proprietary drug-eluting contact lens, is being developed by MediPrint Ophthalmics. In November 2024, the company announced it had completed a Phase 2b Group 1 (low dose) study. Interim analysis showed an about 5.5 mm Hg IOP reduction, with a 14%, 18%, and 19% reduction in IOP each of the 3 weeks, respectively, of the study, according to the company’s press release.
- Mati Therapeutics is developing Evolute, a latanoprost-eluting punctal plug, for multiple ophthalmic conditions, including glaucoma for which it has completed Phase 2 clinical trials.
- In 2025, Amorphex Therapeutics expects to submit an investigational new drug application and begin Phase 2 studies and complete pre-clinical trials for its sustained release prostaglandin and prostaglandin-timolol combination, respectively. The mode of drug delivery being developed is TODDD, which the company describes as a “soft, patented device” worn under the patient’s eyelid.
- Interim analysis presented in October 2024 of a Phase 1/2 study from SpyGlass Pharma, which is developing a bimatoprost-eluting IOL delivered at the time of cataract surgery, showed a mean 43.7% reduction in IOP at month 18 from baseline, and all patients remained off topical IOP-lowering drops at this timepoint. All patients achieved a 20% IOP reduction or more from baseline, and most (96%) maintained an IOP of 18 mm Hg or less through the study.
- PolyActiva is developing the Latanoprost FA SR Ocular Implant, which completed its 48-week Phase 2 clinical trials. During the trials, patients received the first implant at week 0 and a second at week 21; safety and efficacy endpoints were met, according to data presented in October 2024. This implant is fully biodegradable by 4–6 weeks.
- Envisia Therapeutics is developing a travoprost XR sustained release implant (ENV515), which in data reported in 2017 from Phase 2 trials was found to be well tolerated over 11 months. Both the low dose and the high dose demonstrated sustained IOP lowering, with the high dose showing a greater treatment effect.
- Glaukos also has next-generation iDose candidates in the pipeline (iDose TREX and iDose Next Generation), as well as another transdermal sustained-release pharmaceutical platform for glaucoma (GLK-302) in Phase 2 trials.
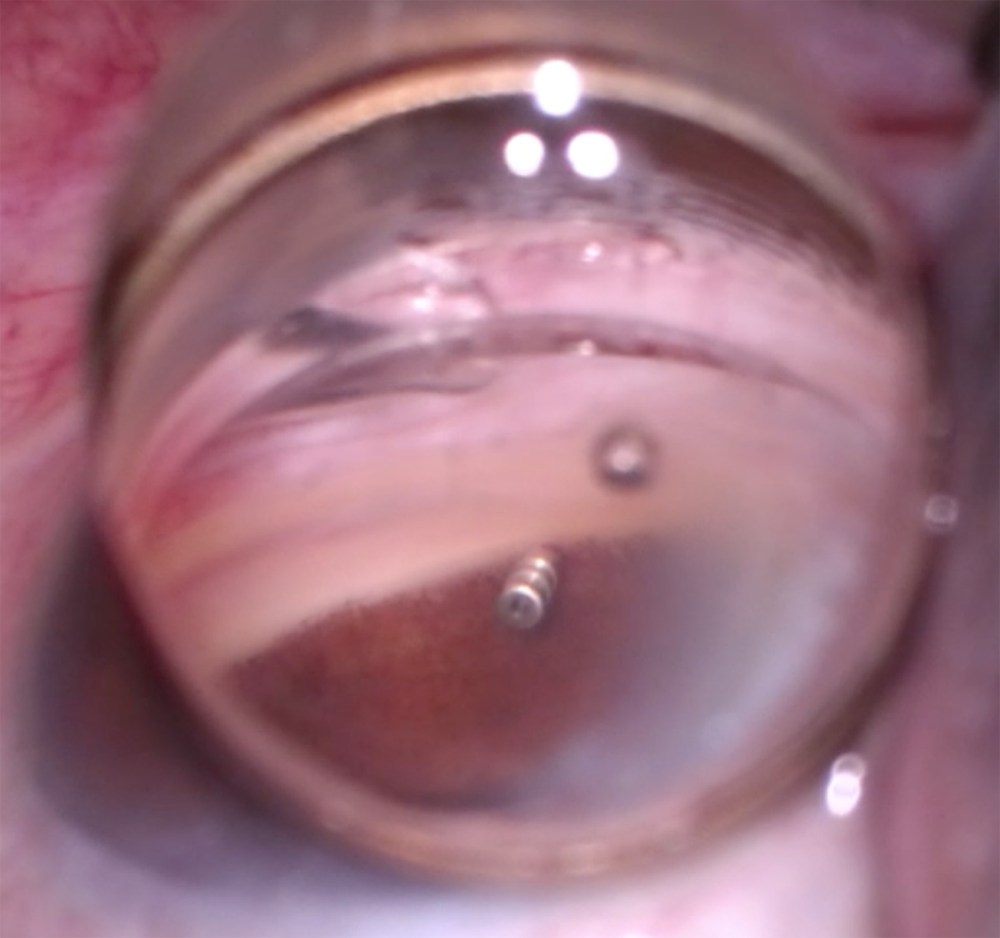
Source: Jacob Brubaker, MD
Takeaways
When it comes to sustained-release options, Dr. Williamson said you have to be honest with yourself and with your patients about the likelihood (or rather the unlikelihood) that they’re taking the medications exactly the way that you’re prescribing them.
“If you know that to be true, you just have to admit that by putting them on a second or third bottle of eye drops, you’re not doing right by the patient. You’re not doing anything wrong, you’re not breaching standard of care, but you’re not doing the best for your patient,” he said. “Do you really trust that they’re going to take that second or third bottle of medication appropriately? Once you admit that they’re not going to be 100% compliant, you have to think, ‘What can I do that is extremely safe but also gives good efficacy and would reduce or eliminate needing to give new medications to my glaucoma patients?’ That’s when you look at the two [sustained-release] options that we have now.”
Dr. Williamson said that Durysta has a short learning curve and can be done in the OR or at the slit lamp. He advised those just starting out to place it in the OR in a pseudophakic patient to gain comfort with the procedure before potentially moving on to slit lamp placement with a phakic patient. For iDose TR, he said if you’ve placed an iStent, especially iStent infinite, then the iDose TR procedure is very similar.
Article Sidebar
Additional thoughts
Ariana Levin, MD, assistant professor, and Albert Bargoud, MD, glaucoma fellow, both in the Department of Ophthalmology at NYU Grossman School of Medicine, New York, New York, also shared their thoughts on drug delivery opportunities for glaucoma.
“Glaucoma requires a lifetime of treatment, so simplifying treatment regimens, reducing side effects, and ensuring affordability are all critical to long-term success,” they said in an email to EyeWorld.
Both said they currently use Durysta, steroid depots like Dextenza (dexamethasone ophthalmic insert, Ocular Therapeutix) and subconjunctival injections, and more recently, iDose.
“The potential benefits for the patient include around-the-clock medication absorption, an improved ocular surface, decreased reliance on patient compliance, and diminished cost of monthly drop bottles. We have had some of our best successes with patients who have mobility issues because drop compliance poses a particular challenge for these patients,” Dr. Levin and Dr. Bargoud said.
They noted that for any intraocular procedure for glaucoma patients, a thorough gonioscopy exam is needed pre-injection, with special attention being paid to the angle anatomy, any signs of uveitis like synechiae, and their preop corneal health.
Looking forward, Dr. Levin and Dr. Bargoud said they are eager for more classes of glaucoma medications to become available in long-acting “dropless” delivery systems. In addition, they said the hope is for longer-lasting depots and potentially refillable delivery systems.
“Delivery systems that adhere to our other commonly used implants, such as intraocular lenses or tube shunts, would also be exciting,” they said.
As a final piece of advice, they noted that intraocular steroid drug delivery is of interest, but it has nuances in the glaucoma population due to the possibility of a steroid-induced IOP spike.
“Glaucoma treatment is an ever-evolving field. Ophthalmologists should update patients on new options. Even patients who have been stable on drops for years might benefit from new options,” Dr. Levin and Dr. Bargoud said.
Relevant disclosures
Bargoud: None
Levin: Alcon
Contact
Bargoud: Albert.Bargoud@nyulangone.org
Levin: Ariana.Levin@nyulangone.org
About the physicians
Jacob Brubaker, MD
Sacramento Eye Consultants
Sacramento, California
Blake Williamson, MD
Williamson Eye Center
Baton Rouge, Louisiana
References
- Berdahl JP, et al. Efficacy and safety of the travoprost intraocular implant in reducing topical IOP-lowering medication burden in patients with open-angle glaucoma or ocular hypertension. Drugs. 2023;84:83–97.
- Medeiros FA, et al. Phase 3, randomized, 20-month study of bimatoprost implant in open-angle glaucoma and ocular hypertension (ARTEMIS 1). Ophthalmology. 2020;127:1627–1641.
- Teymoorian S, et al. Real- world study of the effectiveness and safety of intracameral bimatoprost implant in a clinical setting in the United States. Clin Ophthalmol. 2024;18:187–199.
Relevant disclosures
Brubaker: AbbVie, Alcon, Glaukos
Williamson: AbbVie, Glaukos
Contact
Brubaker: jbrubaker@saceye.com
Williamson: blakewilliamson@weceye.com