Browse articles on EyeWorld.org from the Glaucoma section. EyeWorld is the award-winning member publication of ASCRS. The magazine provides news and updates from the Society as well as clinical features in the areas of cataract, refractive, cornea, glaucoma, and practice management. It publishes quarterly with editorial direction from its medical editorial board and staff.
Glaucoma should not necessarily be an immediate contraindication to premium cataract surgery technologies, such as toric and presbyopia-correcting IOLs and FLACS. Reay Brown, MD, Paul Harasymowycz, MD, and Joey Hsia, MD, said there are situations where these IOL technologies are suited for glaucoma patients, but the decision to offer them is not as straightforward.
Some MIGS options are approved only in conjunction with cataract surgery, while others are not limited in this way. Three ophthalmologists EyeWorld spoke with think there is a value in uncoupling some MIGS procedures from cataract surgery, yet two of the three do not perform off-label standalone MIGS.
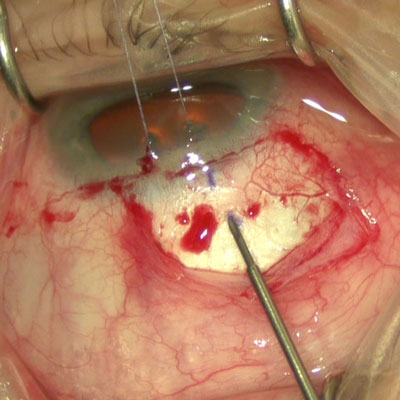
Infection is a risk for any ocular surgery, but there are a few reasons why there isn’t a lot of discussion about infection when it comes to glaucoma procedures. MIGS, which have become increasingly common, have a lower infection risk profile compared to incisional glaucoma surgery.

Nathan Radcliffe, MD
Glaucoma Editor
Glaucoma Editor Nathan Radcliffe, MD, introduces the topics covered in the section, including early experience with Durysta, how other sustained therapies can benefit the glaucoma patient following cataract surgery, and how glaucoma care can be streamlined to keep everyone safe in the pandemic.
Newer steroid and NSAID options (and some off-label administration of antibiotics) that are delivered either via injection or intracanalicular insert can help avoid complicating a patient’s drop regimen that is already subject to non-compliance, instillation issues, potential for ocular surface agitation, and more.
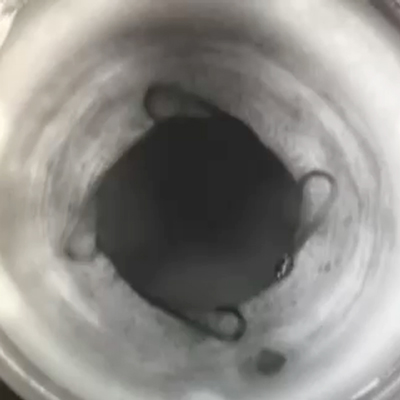
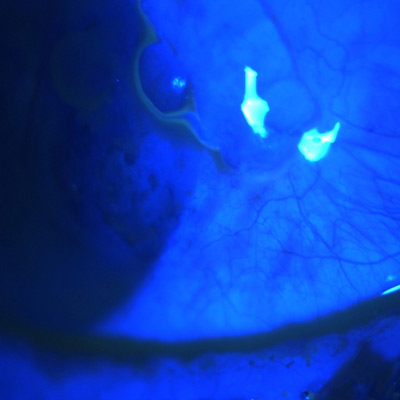
Durysta, the sustained-release bimatoprost implant, 10 mcg (Allergan), has been available with FDA approval in the U.S. market for a year now. EyeWorld spoke with two physicians who started offering it as an option to their patients to hear about their experience thus far.
EyeWorld spoke with several glaucoma specialists who discussed changes they’ve noticed in glaucoma as well as in their own practices due to the COVID-19 pandemic. They shared how they’ve adapted to continue patient care and described the changes they think are here to stay post-pandemic.

Nathan Radcliffe, MD
Glaucoma Editor
As I write this, we are in the ninth month of the COVID pandemic, and each day brings a record number of new cases in the third wave of the virus. As a glaucoma specialist in New York City, my practice has surely seen some ups and downs over the past year. We have had to adapt tremendously, and perhaps now is a good time to reflect on some of these changes.
Where do drops fit in the era of interventional glaucoma—a time when there are new technologies, new drug delivery systems, and several MIGS options?
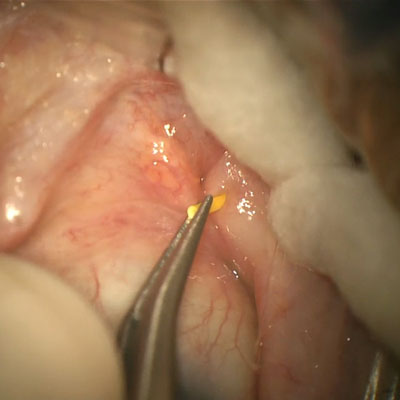
Depending on what you define as a microinvasive glaucoma surgery (MIGS), you might think it first became available several decades ago with endocyclophotocoagulation (ECP), but many define the MIGS era beginning with the iStent (Glaukos), the first ab interno trabecular microbypass device approved by the FDA in 2012.