Glaucoma
Fall 2025
by Liz Hillman
Editorial Co-Director
In the last few issues of EyeWorld, specialists have gone deeper into different types of glaucoma, such as steroid-induced glaucoma and pseudoexfoliation glaucoma. In this issue, Jonathan Eisengart, MD, Davinder Grover, MD, MPH, and Kateki Vinod, MD, discuss pigment dispersion syndrome (PDS) and related pigmentary glaucoma (PG) from diagnosis to treatment and monitoring, and what they want anterior segment surgeons who are not necessarily glaucoma specialists to know about it.
What is PDS and PG?
Dr. Vinod said in an email to EyeWorld that pigment dispersion syndrome is thought to result from posterior iris bowing that causes friction between the iris and the zonules, liberating pigment that is then deposited onto various structures in the eye. Dr. Eisengart said many patients with PDS have concave iris configuration that brings the iris into contact with the zonules.
“This concave iris configuration is thought to be caused by reverse pupillary block in which there is higher pressure anterior to the iris than posterior to it,” he said. “As the iris dilates and constricts in response to changes in ambient light, the posterior iris surface rubs against the zonules and releases pigment. Some people argue that PDS isn’t purely mechanical, and there could be a component of iris pigment epitheliopathy as well.”
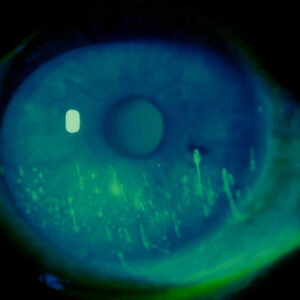
Clinical findings, cited by all the doctors in this article, include pigment on the corneal endothelium (Krukenberg spindle), mid-peripheral iris transillumination defects, and a homogenously and heavily pigmented trabecular meshwork viewed on gonioscopy. Dr. Vinod said that pigment deposition can also occur circumferentially on the posterior lens capsule near zonular attachment. Dr. Eisengart commented that this pigment deposition (called a Scheie stripe) is usually best seen inferiorly. He additionally noted that in light colored eyes a pigment dusting can be seen on the anterior iris surface.
PG is defined by the clinical findings described above in addition to glaucomatous optic neuropathy. Dr. Eisengart said PG is thought to occur due to elevated IOP caused by the pigment cells that clog the trabecular meshwork. However, Dr. Eisengart said he has seen patients with PG who never had an elevated eye pressure recorded in office.
Dr. Vinod said that both PDS and PG are usually bilateral, though they can sometimes be asymmetric. These conditions are most common in younger (30–50 years old), myopic male patients.
“Sometimes, the clinical signs of PDS/PG can be subtle, so it’s essential to have a high degree of vigilance and perform a thorough anterior segment exam, including gonioscopy, in younger patients with high IOP,” Dr. Vinod said. “When assessing a patient for PDS/PG, it’s also important to consider other forms of secondary glaucoma associated with pigment dispersion in the differential diagnosis, such as uveitis-glaucoma-hyphema (UGH) syndrome, uveitis, and intraocular tumors.”
Dr. Grover said the patient presentation for PDS and PG depends on various factors, such as how acute the presentation is, disease stage, and the patient’s healthcare access. “Interestingly, depending on the study, usually around 20–30% of patients with PDS go on to develop pigmentary glaucoma,” he said in an email to EyeWorld. He also said that while the most common patient is a young, white, myopic male, this condition can be seen in all ages and races. “Usually, as the patient ages and their cataracts develop, the lens/zonular complex moves away from the iris and the chance of pigment liberation decreases. However, despite inactive ‘liberation’ of pigment, if a patient has PDS, they have a slightly increased lifelong risk of developing glaucoma, usually 2–4 fold higher than their normal baseline risk.”
Dr. Eisengart said it’s important to distinguish between primary pigment dispersion, which is the focus of this article, and secondary pigment dispersion. The latter occurs post-surgically, often from a sulcus-placed IOL, Dr. Eisengart said, but “anything else that chafes the posterior iris surface, such as a large Soemmering’s ring, can do the same thing.” “Secondary PDS and PG have many of the same findings as primary disease, but it typically occurs in only one eye, and the iris transillumination defects are restricted to areas over the lens implant or other mass in the ciliary sulcus,” Dr. Eisengart said.
Treating and monitoring
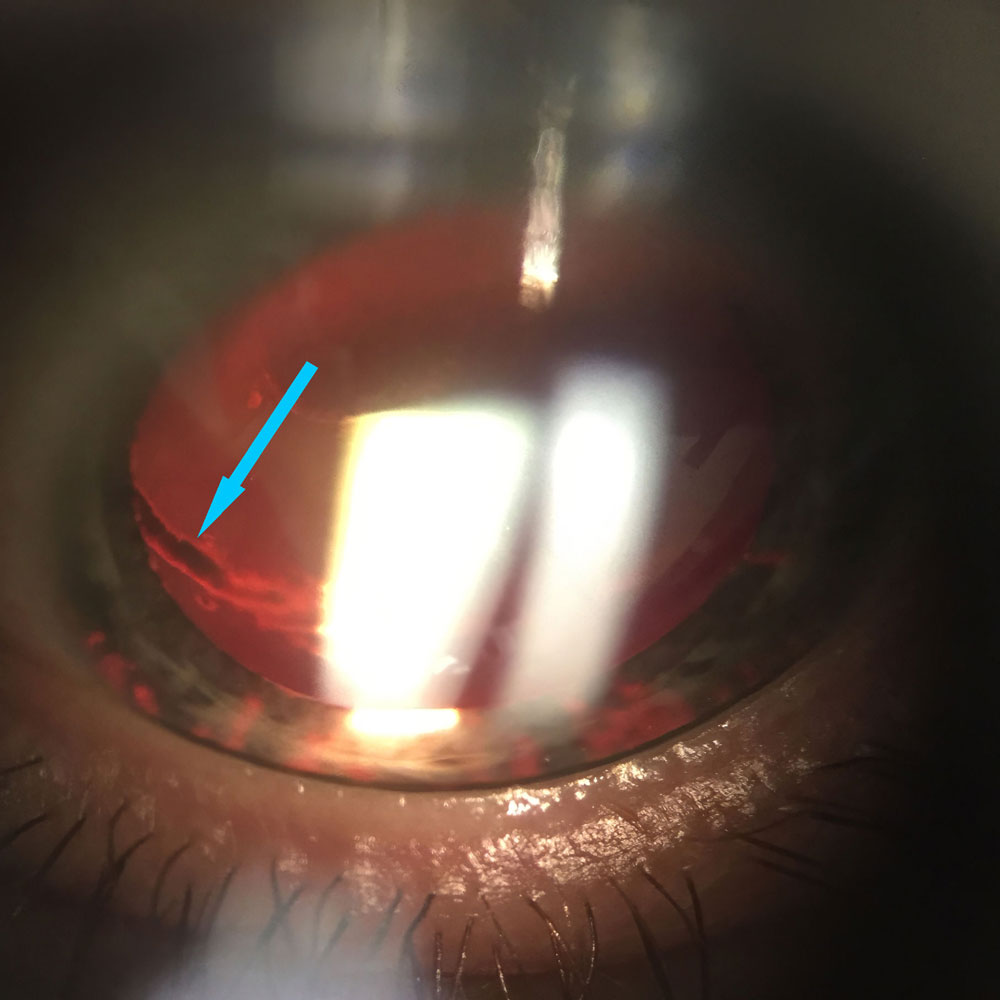
Source: Jonathan Eisengart, MD
Dr. Grover said younger patients are at higher risk for pressure spikes, so he educates them about what to look for and what a spike looks like. Once he establishes a baseline, he thinks following PDS or PG is appropriate.
Dr. Eisengart said if a patient presents with PDS, normal pressure, and a healthy optic nerve, he thinks it’s reasonable to monitor the patient with annual visits, watching for the first signs of glaucoma. Patients with PDS and untreated ocular hypertension or patients with treated and well-controlled PG should be seen every 6 months. Dr. Eisengart also thinks prophylactic treatment for the potential of PG can be prescribed for patients with PDS and ocular hypertension, but he noted that, unfortunately, some eyes aren’t diagnosed with PDS until after glaucomatous damage has occurred.
Dr. Vinod said that the presence or absence of active pigment dispersion, stability of IOP, and OCT RNFL and visual field results influence her monitoring of these patients. “I might follow patients with PDS with normal IOP (or those with ocular hypertension or mild PG that is well controlled on medication) every 6 months, whereas I’ll see patients with active pigment dispersion and refractory IOP elevation far more frequently until we get the IOP under control. Ultimately, the decision of how often to monitor and whether, and how, to treat PDS/PG must be individualized to each patient,” she said.

Source: Jonathan Eisengart, MD
Dr. Vinod mentioned that patients should be counseled about the potential for pigment liberation associated with physical exertion, which can cause IOP spikes presenting as blurry vision and pain. She said that retinal detachment is more common in patients with PDS/PG, so they should be counseled about these symptoms as well.
“For patients whose OCT RNFL or visual fields appear to be progressing despite seemingly well-controlled IOP measurements during office visits, home tonometry is a useful tool to identify IOP spikes related to exercise-induced pigment liberation,” Dr. Vinod added.
As for treatment, Dr. Grover called SLT a “tremendous option.” If that doesn’t safely control IOP, he said IOP-lowering medications are appropriate, as are goniotomy and GATT, if surgery is required. “Some used to advocate for performing a laser peripheral iridotomy in an attempt to resolve the ‘reverse’ pupil block that can sometimes be seen, however, no studies have definitively demonstrated this to be an effective method for treating PDS/PG, and many glaucoma doctors have moved away from this approach,” he said.
Dr. Eisengart acknowledged that some glaucoma specialists would perform a laser peripheral iridotomy, which was intended to eliminate any reverse pupillary block, flatten the iris configuration, and possibly reduce chafing of the iris on the zonules. Dr. Vinod also discussed studies regarding laser iridotomy with the intent to relieve the concave configuration of the iris. She said there is a lack of evidence demonstrating a beneficial effect on visual field progression and the overall disease course.1
Dr. Eisengart noted that treatment of PG is like POAG with a few caveats. When considering laser trabeculoplasty, Dr. Eisengart said eyes with PG are more likely to experience post-laser pressure spikes, due to excess pigment more effectively absorbing the laser energy. “While I typically perform SLT laser 360 degrees in one sitting for eyes with POAG, I am more likely to treat only 180 degrees at a time in eyes with PG to mitigate this risk,” he explained.
Dr. Vinod said due to the risk of IOP spikes post-SLT, she also only initially treats a limited part of the angle (90 degrees) with lower energy settings (starting energy of 0.4 mJ vs. 0.8–1.0 mJ) as a test run. “I check the IOP within an hour after SLT and follow patients with PDS/PG more frequently after the procedure than I would patients with POAG,” she said.
In addition to Dr. Grover, Dr. Eisengart and Dr. Vinod also said that eyes with PG respond well to goniotomy or ab interno trabeculotomy, if surgery is needed. Dr. Vinod said anterior segment surgeons should be aware of IOP spikes that can follow certain MIGS procedures and other complications that can occur with incisional surgery in the PG patient population.
“For example, one retrospective study of patients with OAG undergoing the second-generation iStent inject [Glaukos] found that all 3 patients with PG included in the study developed IOP spikes of 30 mm Hg or higher within the first postoperative month and required subsequent trabeculectomy,”2 she said. “While trabeculectomy remains a good option for those with more advanced PG and refractory IOP elevation, patients with PG who are highly myopic are at higher risk for hypotony-related postoperative complications, such as maculopathy. Glaucoma drainage implant surgery is also a reasonable surgical option for advanced PG.”
PDS long term and final thoughts
PDS can “burn out” over time, usually after a decade, likely due to age-related changes in the lens dimension that relieves friction between the iris and zonules, Dr. Vinod said. This burnout, according to Dr. Eisengart, can also occur because the iris runs out of pigment to release. “The pigment cells on the posterior iris surface do not replace themselves, and once all the cells being chafed are rubbed off, there are simply no more to release,” he said. Whether less chafing is due to lens changes or fewer cells left to chafe, Dr. Eisengart said that IOP can stabilize and become easier to control for these patients.
“Pigmentary glaucoma should remain in the differential diagnosis for an older patient with what appears to be OAG with IOP within the ‘normal’ range (especially if asymmetric) without characteristic findings of PDS/PG, since the IOP can eventually normalize and the degree of pigment deposition in the angle can diminish over time,” Dr. Vinod said. “Classically, older patients with PDS/PG may display the ‘pigment reversal sign,’ whereby the superior angle appears more pigmented due to a decrease in pigmentation in the inferior angle.”
About the physicians
Jonathan Eisengart, MD
Clinical Assistant Professor of Ophthalmology
Cleveland Clinic Cole
Eye Institute
Cleveland, Ohio
Davinder Grover, MD, MPH
Glaucoma Associates of Texas
Dallas-Fort Worth, Texas
Kateki Vinod, MD
Associate Professor of Ophthalmology
Icahn School of Medicine at Mount Sinai
New York Eye and Ear Infirmary of Mount Sinai
New York, New York
References
- Michelessi M, Lindsley K. Peripheral iridotomy for pigmentary glaucoma. Cochrane Database Syst Rev. 2016;2:CD005655.
- Klamann MK, et al. iStent inject in phakic open angle glaucoma. Graefes Arch Clin Exp Ophthalmol. 2015;253:941–947.
Relevant disclosures
Eisengart: Nova Eye
Grover: None
Vinod: None
Contact
Eisengart: EISENGJ@ccf.org
Grover: dgrover@glaucomaassociates.com
Vinod: kate.vinod@gmail.com