Cornea: Beyond the routine
Fall 2025
by Liz Hillman
Editorial Co-Director
Dry eye is not fun for anyone, but Sjögren’s dry eye is serious. This is the message Janet Church, President and CEO of the Sjögren’s Foundation and a patient with Sjögren’s herself, wants to get across to doctors, patients, and the healthcare population at large.
“You have to do something about it and not think of it as ‘just dry eye.’ It’s dry eye and inflammation that is going to progress if you don’t stay on top of it,” she said. “Doctors, if they see dry eye, should be doing a test to see if the patient has Sjögren’s.”

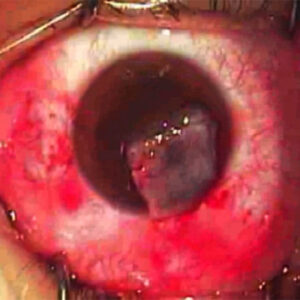
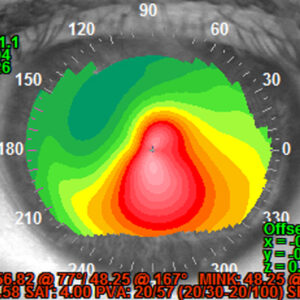
Source: Vatinee Y. Bunya, MD, MSCE
The diagnosis journey
Research suggests that it takes at least 3 years to arrive at a Sjögren’s disease diagnosis from the patient initially presenting with symptoms.1 However, Esen Akpek, MD, said it can take up to 10 years to diagnose a patient with Sjögren’s.
“It’s underestimated and underappreciated,” Dr. Akpek said. “Also, serology, which is the main diagnostic method, is a false negative about 60% of the time in ophthalmology literature. If you look at the rheumatology literature, it’s about 40% of the time. We can safely say that half of the time, serology is a false negative.”
The clinical manifestations of the disease are so diverse that it often takes a multidisciplinary team of physicians to figure out that Sjögren’s is the root cause of the patient’s various symptoms.
Ms. Church said her road to a Sjögren’s diagnosis did not start in an ophthalmologist’s office. She noticed she was getting thirstier (she later learned that a human can’t discern their mouth is dry until 50% of their salivary function is already gone). Reflecting back after her diagnosis, Ms. Church said she had sinus infections and lymph node and parotid gland swelling throughout her life. She hadn’t had any discernible eye dryness prior to her diagnosis (though she said she does now) but noted that it’s hard to determine dryness as a patient because dryness, to an extent, is normal to the patient; it’s all they know. “The patient isn’t always the one coming to the doctor and saying, ‘I’m super dry; something’s wrong,’” she said. Often a patient first comes to a doctor’s office with unexplained fatigue, not recognizing (or not having) signs of dryness.
Ms. Church was hospitalized for a month with neurological issues (aseptic meningitis) at 40 years old. Nine months later at age 41, she was finally diagnosed with Sjögren’s disease. Neurological symptoms are common to Sjogren’s patients, though aseptic meningitis is less common.
About a year after her diagnosis, Ms. Church said she started feeling eye sensitivities and began using drops. Now, she has severe dry eye that has aggressively progressed despite treatment.
Vatinee Y. Bunya, MD, MSCE, said that she uses the 2016 American College of Rheumatology-European League Against Rheumatism (ACR-EULAR) classification criteria to diagnose Sjögren’s disease. “There are currently no diagnostic criteria for Sjögren’s disease, so clinicians are using these classification criteria to identify Sjögren’s patients,” she said, including the below ACR-EULAR criteria2:
- Anti-SSA/Ro antibody positivity (3 points)
- Positive lip biopsy (focal lymphocytic sialadenitis with a focus score of ≥1 foci/4 mm2) (3 points)
- Abnormal ocular staining score of ≥5 (or a van Bijsterveld score ≥4) (1 point)
- Schirmer test without anesthesia ≤5 mm/5 minutes (1 point)
- Unstimulated salivary flow rate of ≤0.1 mL/minute (1 point)
She said individuals who meet eligibility criteria for Sjögren’s have a score of 4 or more. “Of note, the two ocular tests that are included in the criteria include an abnormal ocular staining score or an abnormal Schirmer test without anesthesia,” she said. “Therefore, it’s important for ophthalmologists to perform these two tests in any dry eye patient who may have Sjögren’s disease.”
Dr. Bunya also said that while there are newer diagnostic tests for dry eye disease, none have been shown to distinguish dry eye that is associated with Sjögren’s. “Ophthalmologists and optometrists play a critical role in diagnosing Sjögren’s patients early on in their disease, as many studies have shown that dry eye is one of the most common symptoms of Sjögren’s disease,” Dr. Bunya said. “I recommend that the ocular staining score and Schirmer test without anesthesia be performed on all new dry eye patients. Dry eye patients should be referred for systemic workups for Sjögren’s disease if they meet any of the ocular criteria, if they have a positive review of systems for symptoms such as joint pain, dry mouth, or fatigue, or if they have dry eye disease that is refractory to treatment.”
Once a patient has met the eligibility criteria for Sjögren’s, Dr. Akpek said the patient should have a battery of serology tests that include testing for Sjögren’s or another underlying autoimmune disease.
Dr. Akpek said the gold standard for diagnosis is a minor salivary gland biopsy from the inner side of the lower lip, but this is invasive and can be difficult to perform accurately.
An area of research for a different form of Sjögren’s diagnosis is tear film cytokines and chemokines, Dr. Akpek said. She mentioned the research from Sharmila Masli, PhD, at Boston University, whose lab centers around tear film cytokines, chemokines, and different types of mucins to differentiate Sjögren’s from non-Sjögren’s dry eye.
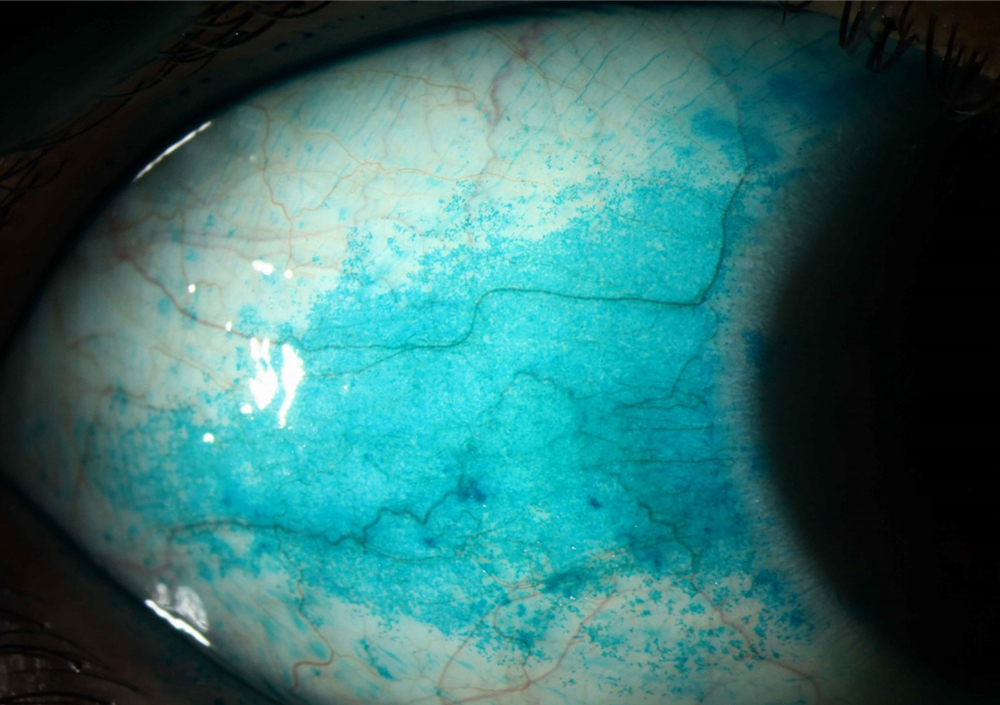
Source: Vatinee Y. Bunya, MD
Available treatments and future hopes
Dr. Bunya said she has a lower threshold for starting prescription dry eye treatments in patients with Sjögren’s than non-Sjögren’s dry eye patients, including topical cyclosporine and lifitegrast. “If a patient has concurrent blepharitis and/or meibomian gland dysfunction, I will start them on treatment for those diseases as well,” Dr. Bunya said, adding, “Sjögren’s patients with low aqueous tear production may benefit from punctal occlusion or cautery, as well as autologous serum drops earlier than non-Sjögren’s patients. Finally, for moderate to severe Sjögren’s-related dry eye disease, scleral lenses can be helpful. If a Sjögren’s patient’s ocular surface disease cannot be controlled with local measures or treatments, I will work with his or her rheumatologist to start or modify their systemic treatments. Sjögren’s patients often require frequent follow-up and a combination of therapies to control their ocular surface disease.”
Dr. Akpek said if patients are diagnosed at an earlier stage, topical and systemic anti-inflammatories, such as steroids, topical lifitegrast, and topical cyclosporine, can be helpful. For patients who are more advanced, while it’s not too late to start, the efficacy of these options might not be as good. An anti-inflammatory that’s not yet approved is ILYX-002 (Iolyx Therapeutics). In May 2025, Iolyx Therapeutics announced positive Phase 2 topline data for the topical immunomodulator specifically targeting autoimmune dry eye and ongoing Phase 3 study design, which is expected to begin enrolling this year.
“It’s good news that there’s at least one company that is working on this and that their results seem extremely promising. It is important that we develop specific treatments for Sjögren’s-related dry eye,” Dr. Akpek said.
Ms. Church tried but could not tolerate topical cyclosporine or lifitegrast. She has had a positive experience with hydroxychloroquine for fatigue symptoms.
Dr. Bunya said there are several investigational treatments in Phase 3 clinical trials or with recent FDA approval that could be helpful for Sjögren’s dry eye. “For example, acoltremon [Tryptyr, Alcon], a first-in-class transient receptor potential melastatin 8 (TRPM8) receptor agonist, was recently approved by the FDA for the treatment of dry eye disease,” she said. “In addition, Stuart Therapeutics announced the initial results of its Phase 3 clinical trial evaluating ST-100 (vezocolmitide). The company reported that vezocolmitide is part of a novel drug class of collagen mimetic peptides and works by targeting and repairing damaged collagen. However, further larger studies are needed.”
Cenegermin (Oxervate, Dompe) is under investigation for severe Sjögren’s-related dry eye. Dr. Akpek said this is an interesting option because it’s an indirect mechanism of action to reverse the damage to the sub-basal corneal nerve plexus and corneal sensation for these patients. Lacripep (Tear Solutions) was also investigated for patients with primary Sjögren’s, demonstrating improvements in signs and symptoms that will be used as areas for future studies.3
As a final message to ophthalmologists, from a Sjögren’s patient and Sjögren’s patient advocate, Ms. Church said that these patients can go blind from this disease and “the way to blindness is pretty painful.” She said cataract surgeons should also understand that improving dryness for a Sjögren’s patient prior to surgery should be a priority, and to remember that healing for these patients is slower.
“The thing I want doctors to understand is this dry eye is not your typical dry eye, and it needs to be diagnosed; patients need to be on treatment and more than just over-the-counter eye drops. They should be on treatment with lifitegrast, cyclosporine, or whatever is coming … but they should also be talking with their doctor about systemic therapy such as an immunomodulator (like hydroxychloroquine) or anti- inflammatory depending on their symptoms.”
She said that more studies are needed to better understand the disease and how it shows up differently in people. “It can attack anyone at any age, and doctors need to include Sjögren’s in their differential diagnosis when they see unusual fatigue, dryness, or neurological symptoms,” Ms. Church said.
Dr. Akpek agreed that heightened awareness is needed in the field, in addition to development of point-of-care testing for Sjögren’s biomarkers in the tear film. She said ideally, a systemic treatment targeted based on expression profiles of cytokines and chemokines is needed as well.
Dr. Bunya said her wish list is for better screening tests, treatments that slow or stop the disease’s progression, and eye drops that specifically target the underlying pathophysiology that causes ocular surface disease in Sjögren’s patients.
About the sources
Esen Akpek, MD
Bendann Family Professor of Ophthalmology
Wilmer Eye Institute
Johns Hopkins University School of Medicine
Baltimore, Maryland
Vatinee Y. Bunya, MD, MSCE
Harold G. Scheie Chair and Associate Professor of Ophthalmology
Scheie Eye Institute
University of Pennsylvania
Philadelphia, Pennsylvania
Janet Church
President and Chief Executive Officer
Sjögren’s Foundation
Reston, Virginia
References
- Sjögren’s Foundation. sjogrens.org/understanding-sjogrens/diagnosis. Accessed July 1, 2025.
- Shiboski CH, et al. 2016 American College of Rheumatology/European League Against Rheumatism classification criteria for primary Sjögren’s syndrome: a consensus and data-driven methodology involving three international patient cohorts. Ann Rheum Dis. 2017;76:9–16.
- Tauber J, et al. Lacripep for the treatment of primary Sjögren-associated ocular surface disease: results of the first-in-human study. Cornea. 2023;42:847–857.
Relevant disclosures
Akpek: None
Bunya: Kowa
Church: None
Contact
Akpek: esakpek@jhmi.edu
Bunya: Vatinee.Bunya@pennmedicine.upenn.edu
Church: jchurch@sjogrens.org