Cornea
Spring 2025
by Ellen Stodola
Editorial Co-Director
BrightMEM corneal allograft (Brightstar Therapeutics) is a new treatment option for ocular surface disease. Several sources discussed BrightMEM, their experience using it, and its treatment applications.
Albert Cheung, MD, said that BrightMEM is a decellularized Descemet’s membrane product that is used for its inherent properties in cases of ocular surface disease. The anterior aspect of Descemet’s membrane contains proliferative proteins that are similar to those found in the limbal basement membrane, he said, adding that these are not typically present in the central cornea.
Source: Joshua Hou, MD
Pre-clinical studies have demonstrated the ability of Descemet’s membrane to act as a substrate for epithelial cells and limbal stem cells, to be more resistant to degradation than amniotic membrane, and to maintain greater transparency than amniotic membrane, Dr. Cheung said.
Joseph Tauber, MD, said the term “BrightMEM” is used for both a product and a procedure, BrightMEM anterior keratoplasty (BMAK), in a unique way. “Descemet’s membrane is the back layer of the cornea, and we’re using it on the front of the eye, and we’re doing that because of some recently discovered science from Joshua Hou, MD, at the University of Minnesota, who found that the anterior side of Descemet’s membrane, the fetal banded portion of Descemet’s membrane, contains proteins that are very supportive, encouraging of stem cells,” he said.
Why does that matter? “You have to understand the normal healing process,” he said, “where there are undifferentiated stem cells at the limbus. Something happens in the central cornea, there’s a signal to heal, and the cells march their way across the cornea. They leave the niche, they lose their supportive protein microenvironment, and they start to differentiate, and hopefully grow back over the entire surface. If those cells could grow over a membrane that maintains that regenerative capability, that would be a good thing,” Dr. Tauber said.
“The original development concept was to use that membrane, grow a layer of cells on it, and provide surgeons with both membrane and cells,” he said. “That is a more challenging regulatory pathway for FDA approval that’s on our radar. … But it turned out that the decellularized Descemet’s membrane is wonderful just by itself.”
Rajan Shukla, CEO of Brightstar Therapeutics, has worked in front-of-the-eye therapeutics since 2014 and has previously worked with serum tears. It was this work and his interest in novel therapies for severe dry eye and ocular surface disease that ultimately led him to BrightMEM.
“When you look at quality of life, severe dry eye disease is comparable to hip fracture and severe angina in that it impedes patients from living their lives. In many cases, patients can suffer from both significant vision loss and constant eye pain,” he said.
BrightMEM helps fill a niche intended for these severe ocular surface disease patients who have impaired healing of their cornea. “All of the limbal stem cells, all of the cells in the eye that produce the clear cornea [epithelium], live in a specialized area called the limbal niche, which is in a ring around the cornea,” Mr. Shukla said. “These cells produce the corneal epithelium, and when the cornea is damaged or the epithelium is removed, the limbal stem cells migrate out of the niche and start repopulating the cornea, but once they leave that niche, they lose their stem cell phenotype. What that means is that stem cells are capable of self-renewing so they can do horizontal division, making another stem cell, or they can differentiate into mature epithelial cells, which form the clear cornea. But once they leave the niche, they lose that ability, so what we saw [is] if you’ve lost enough stem cells, they can’t do that double duty of maintaining their own population and healing the cornea. That’s when you start to see… conjunctiva growing over [the cornea], and that ends up causing pain and photophobia. So patients are in a terrible condition where they can’t see and they’re in pain all the time.”
The company was looking for a new membrane that was both durable and optically clear, so it can act like the limbal niche and maintain the stem cells long term.
Mr. Shukla said BrightMEM is designed to be a long-term solution for patients. It’s designed to take advantage of Descemet’s membrane’s resistance to degradation despite breakdown of other parts of the cornea. The other major discovery, he said, is that this membrane has proteins that are found in the limbal niche but not found in the central cornea so these can act like a limbal niche, ideally keeping stem cells alive long term. He said laboratory data has demonstrated this, and it has been compared to amniotic membrane. “You can grow limbal stem cells on BrightMEM, and they form a stratified layer of cornea, with layers of mature epithelium, just like the normal eye would, but the very bottom layer still expresses the markers for limbal stem cells, so we’re creating this reservoir on the ocular surface of stem cells,” Mr. Shukla said. For patients with persistent epithelial defects, there is an overwhelmed stem cell population and a sick ocular surface because the limbal stem cells can’t keep up with maintaining the clear epithelium.
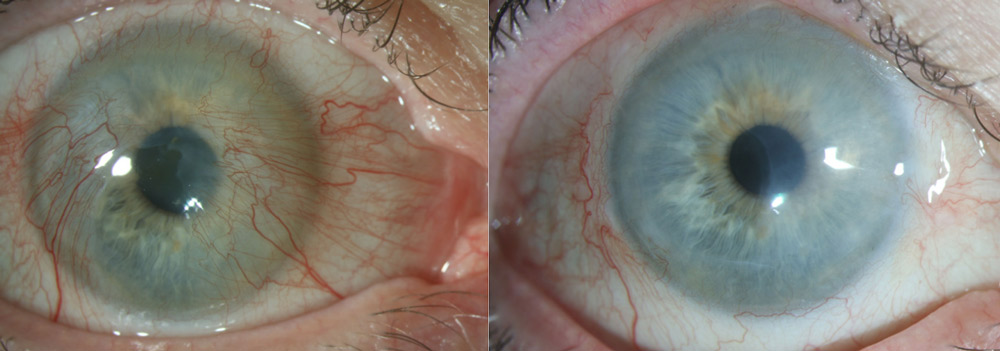
Mr. Shukla mentioned a pilot clinical study in humans that, to date, has had 14 completed cases. In that study, patients with congenital aniridia, a genetic disorder of the eye associated with limbal stem cell deficiency, were enrolled for treatment. Fifty-seven percent of patients had neovascularization in the central cornea degrading their vision. All patients underwent transplantation of a BrightMEM allograft. The intention was to evaluate whether BrightMEM could augment the remaining limbal stem cell population on the ocular surface in these patients, prevent neovascularization, and rehabilitate vision long term. Interim results at 6 months were favorable, with all patients healed and no recurrence of central neovascularization. In addition, all of the patients who were candidates wanted to have their second eye done.
Mr. Shukla also discussed how the product could be used for ocular pain, as it has the potential to provide immediate pain relief from postop day 1. The thought of using it for this application came about when BrightMEM was given to a patient who had ocular toxicity associated with a new cancer drug for end-stage cervical cancer. The pain from the ocular toxicity was debilitating, and with limited options, the surgeon used BrightMEM. Within days after the procedure in her first eye, the pain was completely gone. The surgeon soon treated the second eye in the same way with similar results.
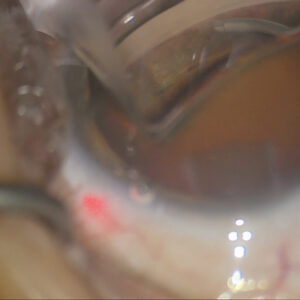
According to Dr. Cheung, the BrightMEM procedure initially involves a keratectomy where irregular epithelium and anterior corneal scarring is removed from the cornea to create a bed for the BrightMEM. The BrightMEM is delivered to the surgeon as a modified, decellularized, pre-stripped, pre-stained, and pre-stamped Descemet’s membrane that is partially attached to its donor cornea. The donor corneal button is punched to size and placed on the dried anterior surface of the patient’s cornea. The interface fluid is wicked away for 1–2 minutes. Afterward, the anterior stromal button is removed, and the Descemet’s membrane (BrightMEM) is left behind. This is smoothed out and allowed to dry. If there are folds or wrinkles, adding a small amount of balanced salt solution will allow those to flatten. The physician then squeegees the interface fluid out to completely smooth the BrightMEM on the corneal surface. Fibrin glue is applied to seal the edges, and a bandage contact lens is placed over the eye. The entire procedure takes 15–20 minutes and can be performed under topical or block anesthesia, Dr. Cheung said.
Dr. Tauber said the procedure is easy to learn. Most surgeons know how to perform a superficial keratectomy, to remove scar and vessels in order to expose a bare surface for BrightMEM placement.
If there’s a new thing for surgeons to learn, Dr. Tauber said it’s the maneuvers to straighten and flatten the tissue because it’s so thin (15 microns). It wrinkles easily, and it doesn’t take much of a touch to produce another wrinkle. There are some specific maneuvers to get it to lay down flat, but if you do a typical case without a lot of scarring, it’s a 15-minute case under topical anesthesia. More extensive scarring can lengthen the case significantly.
Dr. Cheung said the main indication has been to heal persistent epithelial defects from a variety of etiologies. It is also currently being studied in cases of partial limbal stem cell deficiency, and Dr. Hou originally developed it with partial stem cell deficiency in mind. Dr. Tauber said that the labels characterizing superficial corneal disease get a bit confusing and many patients who fall under these indications have overlapping pathology with other epithelial diseases, so there may be other corneal conditions that can benefit from BrightMEM. For example, Dr. Tauber said many patients who have received BrightMEM had persistent epithelial defects and ocular pain from partial limbal stem cell deficiency but also had some degree of neurotrophic keratitis. As such, surgeons may find it helpful in a variety of ocular surface disease patients. However, Dr. Tauber cautioned physicians looking to use BrightMEM should take care to avoid cases with 100% stem cell deficiency, particularly as their first cases.
Dr. Cheung said BrightMEM offers the benefit of improving epithelialization by giving epithelium a substrate to grow over. This could be compared to using amniotic membrane as an onlay, however, amniotic membrane doesn’t last as long, and it is relatively opaque.
Mr. Shukla said BrightMEM is commercially available. As of late 2024, around 60 surgeons have used BrightMEM. “We expect this to grow substantially through 2025,” he said.
Discussing some of the clinical data so far, Dr. Tauber said there has been data accumulated on about 85 cases. “We’re seeing about 90% fully healed at a mean of 32 days with a graft retention rate over 90%. Durability has been excellent, with 91% still healed at 6 months and 100% at 12 months. Healing rates vary somewhat, depending on the subgroup,” Dr. Tauber said, again stressing that it’s important for surgeons to pick appropriate cases.
BrightMEM can also be an adjunctive treatment used in combination, Dr. Tauber said. If you put down the ideal substrate, and you were to flood the surface with serum tears full of growth factors, that’s a good combination for patients who have very few stem cells. If you add stem cells, that’s even better. “I see collaboration as the future with what we’re doing. This is not necessarily a standalone procedure,” he said.
Dr. Tauber said this is a new and different concept for treating ocular surface disease. It adds a different mechanism for improving corneal epithelial health that is separate from current therapies that focus mainly on improving the tear film and lubricating the cornea. “We have many anti-inflammatories and aqueous or water substitutes [for tears]. We also have a kind of lipid substitute, something to prevent evaporation [of tears].” Now, with BrightMEM, there is a substrate to promote the healing of the epithelium. “I think the new frontier is growth factor supplementation,” he said, adding that a number of companies are also exploring this space.
About the sources
Albert Cheung, MD
Virginia Eye Consultants
Norfolk, Virginia
Rajan Shukla
Chief Executive Officer
Brightstar Therapeutics
Lexington, Kentucky
Joseph Tauber, MD
Chief Medical Officer
Brightstar Therapeutics
Leawood, Kansas
Relevant disclosures
Cheung: Brightstar Therapeutics
Shukla: Brightstar Therapeutics
Tauber: Brightstar Therapeutics
Contact
Cheung: ACheung@cvphealth.com
Shukla: rajan@brightstartx.com
Tauber: jtauber@brightstartx.com