Cataract: Complicated cases
December 2021
by Ellen Stodola
Editorial Co-Director
Negative dysphotopsia is an issue that patients may face following cataract surgery. While it frequently resolves on its own, several physicians said it’s important to discuss it with patients and explain what’s going on.
Jack Holladay, MD, highlighted risk factors that make a patient more likely to develop negative dysphotopsia, though he said you can’t definitively predict who will experience the complication. There are risk factors that can predispose it, but it’s difficult to predict when it will happen and to whom.
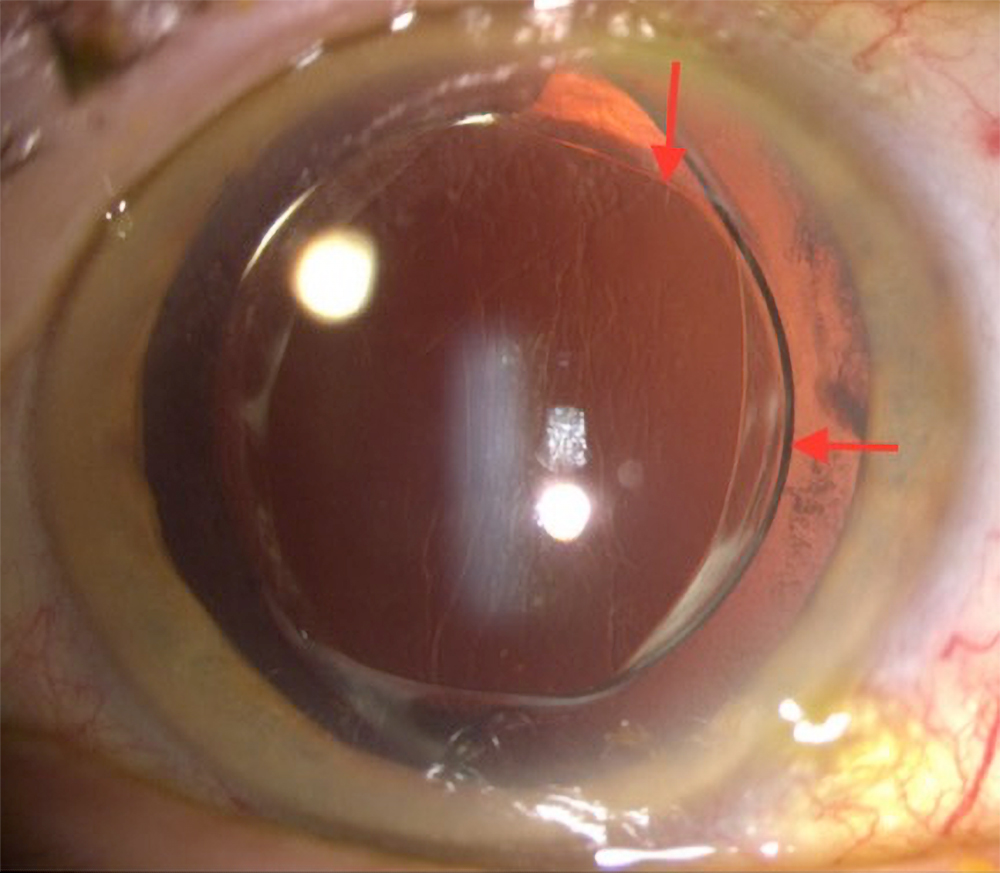
Source: Nicole Fram, MD
Dr. Holladay noted primary risk factors from a paper he wrote,1 including a smaller photopic pupil, larger positive angle kappa, the shape of the IOL, smaller axial distance of the IOL behind the iris, nasal anterior capsule overlying anterior nasal IOL, higher dioptric power if equi-biconvex or plano-convex, and if the optic-haptic junction of the IOL is not horizontal. Secondary risk factors include the edge design of the IOL (if it’s truncated vs. rounded), the material of the IOL, and negative aspheric surfaces.
Patients who have a small pupil (around 1.5–2 mm) are at higher risk for experiencing negative dysphotopsia when in bright light, Dr. Holladay said.
Dr. Holladay said he tries to stress to physicians how important these identifying measures are in helping to recognize high-risk patients preoperatively.
Negative dysphotopsia is caused by a gap in rays that pass through the lens that miss the lens. There’s not a gap before surgery with the natural crystalline lens, Dr. Holladay said. “There are no rays that make it into the eye that can get between the crystalline lens and the iris,” Dr. Holladay explained. “That gap appears as a dark crescent or circle where the rays are missing on the retina.
“What we showed in our paper is as you adjust the lens and make that gap bigger and various things in the eye different, you can make that crescent move more temporally, and you can make the thickness wider or thinner by how much larger that gap is between the rays that miss the lens and the rays that pass through the lens,” he said.1
Nicole Fram, MD, noted that this is a complication that can happen with any type of lens. The patient often has had perfect cataract surgery and is seeing 20/20. However, they may see a dark shadow off to the side temporally.
“The first thing you want to do is reassure the patient that you know what it is,” she said, adding that it’s also important to rule out any retinal or neurological pathology such as a retinal tear or detachment.
Dr. Fram said Robert Osher, MD, reported that the percentage of patients with negative dysphotopsia on postop day 1 is approximately 15%.2 The working theory of negative dysphotopsia based on ray tracing is that some light rays are bent by the lens and some are missed,3 which creates an illumination gap on the nasal retina causing a dark shadow temporally.
In talking to patients before surgery, Dr. Fram said that she mentions that this is something that can occur, particularly in patients needing a high power IOL or who have a large angle kappa. However, she doesn’t mention the word “dysphotopsia.” Instead, she explains that the intraocular lenses used have the potential to cause glare or shadowing, and if this a problem, she can help.
Dr. Fram said it’s important to ask if this issue is always there or it comes and goes. “If it’s not always there or only there in certain lighting, I’m optimistic that the patient will adapt and not require an intervention,” she said. Dr. Osher’s research found that 97% of cases resolved within a year.2 That’s very encouraging to the patient, she said.
Randall Olson, MD, agreed that this is an issue that’s often seen in the first couple of weeks after surgery, though he also sees a lot of patients who have been referred to him with a persistent problem.
For the majority of patients, it’s not a big issue. However, Dr. Olson stressed that it’s important not to tell patients that you don’t know what it is or that it’s a rare complication. “I let them know ahead of time that these IOLs are smaller than our own lens, things can happen, but these things are normal,” he said.
“Most of the patients I see, by the time they get to me, have been told they should be happy with their vision, and it’s an uncommon complication,” he said. “The angriest are those who have been led to think they’re crazy.” Dr. Olson said he tells patients that this is something that will go away on its own.
The hard part with negative dysphotopsia, Dr. Fram said, is explaining to patients that it’s unknown whether or not it will happen in the other eye. Some patients are cautious about moving on to the other eye, and that’s where you get into a predicament, she said. “If you have a big difference between the eyes or anisometropia, you want to move on to the other eye.”
There are a couple of ways to approach this. The physician could say, “We don’t know the true incidence, but there’s about a 50% chance that this could happen in the other eye,” Dr. Fram said. Assure the patient that if this happens, you can help them. You can wait a month to see if it starts to get better or choose a different strategy for the other eye, which is to put the lens in a different position. This involves doing a primary reverse optic capture (ROC) so that the optic is on top of the capsule with the haptics in the capsule bag oriented vertically. This is best performed with a 3-piece IOL. “We prefer the L161AO SofPort IOL B and L [Bausch + Lomb] as silicone has a lower index of refraction than acrylic and is more friendly in the sulcus,” Dr. Fram said. A single-piece acrylic in the primary ROC position can lead to capsule block and is not ideal for this procedure, she said. Samuel Masket, MD, et al.4 reported on this and found that 100% of patients did not have negative dysphotopsia in the second eye when this strategy was used.
That is for the patients who really can’t wait and are bothered by the first eye but need to move on to the other eye because they’re not functional, Dr. Fram said. But for many patients, she’s able to wait and see if the issue resolves over time. In her experience, typically by 3 months, the patient has improved.
For patients in whom the negative dysphotopsia has persisted for 6 months or longer, Dr. Fram said you may need to move on to other options. The treatment strategy is to move the optic forward and thus move the illumination gap outside of the nasal retina. The nasal capsule has also been implicated in the multifactorial etiology of negative dysphotopsia, and covering the nasal capsule with the optic has improved symptoms. Strategies for treatment in persistent negative dysphotopsia include ROC, sulcus IOL, piggyback IOL, and nasal capsulectomy. She said secondary ROC works best if the patient’s capsulotomy is 4.5–5 mm and if they have an AcrySof IOL (Alcon) with the haptics oriented vertically.4,5 It is less predictable with other platforms as they are more rigid and may slip back into the bag, she said. If ROC is not possible, the physician can perform an IOL exchange where a 3-piece lens is placed, and ROC is done.
“If the anterior capsulotomy won’t allow for that, we put the lens in the sulcus,” Dr. Fram said. With this option, she will fixate the lens to the iris gently, so it doesn’t move over time. In her research with Dr. Masket,5 ROC worked 96% of the time, and sulcus placement worked 86% of the time. Patients should understand that after ROC procedures early fibrosis of the capsule may occur, requiring a YAG posterior capsulotomy.
Piggyback lenses can also be used, as can a secondary IOL on top of the lens to help scatter light, which worked 73% of the time. Bag-to-bag exchange has not worked well in Dr. Fram’s experience.
Dr. Fram also mentioned a strategy reported by Folden6 and Cooke7 of using nasal capsulectomy, and her earlier research with Dr. Masket also indicated that nasal capsule was implicated in the etiology of negative dysphotopsia. However, many of the strategies such as ROC and sulcus placement move the optic forward and may fit with the ray tracing theory as well. Nasal capsulectomy has reduced rather that cured negative dysphotopsia in Dr. Fram’s experience. This may be a good strategy for a patient with a toric lens with the haptics oriented horizontally where you do not want to exchange the IOL and cannot ROC that particular IOL.
Dr. Olson said that he tells patients to wait at least 6 months to give the brain time to adapt before pursuing any surgical options.
He said that options for eliminating the capsule on the nasal side or moving the optic to the top of the capsule are both effective options. A piggyback can be effective, but you have to make sure you have plenty of room so you don’t get pigment dispersion.
The key in these cases, Dr. Olson said, is to not think of negative dysphotopsia as “abnormal.” It’s part of what lenses do.
Dr. Olson concluded by stressing several key principles. Let patients know ahead of time that this is common and normal. Physicians should be able to recognize what it is so they can help the patient. Dr. Olson also tells patients, “The more you’re concerned about it, the harder it is for the brain to ignore.” He tries to get patients to not worry too much about it because it will either resolve or can be addressed if it persists.
About the physicians
Nicole Fram, MD
Advanced Vision Care
Los Angeles, California
Jack Holladay, MD, FACS
Clinical Professor
Department of Ophthalmology
Baylor College of Medicine
Houston, Texas
Randall Olson, MD
Chair
Department of Ophthalmology and Visual Sciences
John A. Moran Eye Center
University of Utah
Salt Lake City, Utah
References
- Holladay JT, Simpson MJ. Negative dysphotopsia: Causes and rationale for prevention and treatment. J Cataract Refract Surg. 2017;43:263–275.
- Osher RH. Negative dysphotopsia: long-term study and possible explanation for transient symptoms. J Cataract Refract Surg. 2008;34:1699–1707.
- Coroneo MT, et al. Off-axis edge glare in pseudophakic dysphotopsia. J Cataract Refract Surg. 2003;29:1969–1973.
- Masket S, et al. Surgical management of negative dysphotopsia. J Cataract Refract Surg. 2018;44:6–16.
- Masket S, Fram N. Pseudophakic negative dysphotopsia: Surgical management and new theory of etiology. J Cataract Refract Surg. 2011;37:1199–1207.
- Folden DV. Neodymium:YAG laser anterior capsulectomy: surgical option in the management of negative dysphotopsia. J Cataract Refract Surg. 2013; 39:1110–1115.
- Cooke DL, et al. Resolution of negative dysphotopsia after laser anterior capsulotomy. J Cataract Refract Surg. 2013;39:1107–1109.
Relevant disclosures
Fram: None
Holladay: None
Olson: Perfect Lens, Perceive Bio, TMClear
Contact
Fram: drfram@avceye.com
Holladay: holladay@docholladay.com
Olson: RandallJ.Olson@hsc.utah.edu