Browse articles from EyeWorld.org related to dry eye. EyeWorld is the award-winning member publication of ASCRS. The magazine provides news and updates from the Society as well as clinical features in the areas of cataract, refractive, cornea, glaucoma, and practice management. It publishes quarterly with editorial direction from its medical editorial board and staff.
➤ FDA accepts NDA for ‘first-of-its-kind’ anti-inflammatory for dry eye
➤ Positive Phase 3 results for investigational presbyopia therapy
➤ Study: long-term visual outcomes with remote monitoring technology
➤ Companies partner to create tool to assess fall risk
➤ ASCRS news and events
➤ ‘Visionary Report’ sheds light on understanding, perspectives of AMD
➤ NDA submitted for Demodex blepharitis treatment
➤ Study complete for trial of oral diabetic retinopathy therapy
➤ Enrollment complete for study comparing dry eye therapeutic device to cyclosporine
➤ Gene therapy developed for ciliopathy associated with a form of Leber congenital amaurosis
➤ ASCRS news and events
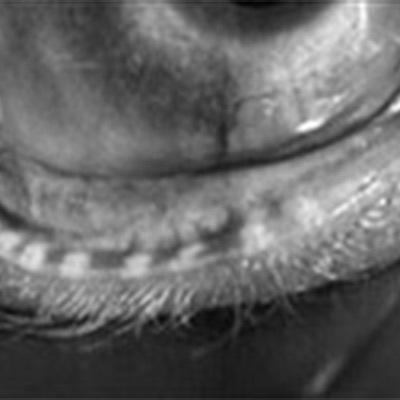
Aqueous tear deficient dry eye and evaporative dry eye—distinguishing between the two, knowing when both are at play, and treatment options are of growing importance as more patients are identified with some form of dry eye and as certain refractive procedures depend on a healthy ocular surface.
➤ New device for bladeless goniotomy
➤ FDA approves the first at-home digital vision test
➤ Alcon to acquire Aerie Pharmaceuticals
➤ Study: Phase 2 trial for investigational dry eye therapy meets primary endpoint
➤ ASCRS news and events
➤ FDA approves new multifocal IOL
➤ FDA approves new biosimilar
➤ First patient dosed in Phase 3 trial for investigational dry eye drug
➤ First patient dosed in Phase 2 trial investigating sustained delivery for wet AMD
➤ First patient dosed in Phase 2 study of small molecule to treat diabetic retinopathy
➤ Enrollment complete in Phase 3 trials evaluating myopia progression therapy
➤ Study emphasizes need for earlier glaucoma screening in black patients
➤ Company acquires diagnostic technology
➤ ASCRS news and events
➤ Small-aperture IOL receives FDA approval
➤ First patient enrolled in new LASIK trial
➤ Safety of sustained-release intravitreal implant confirmed
➤ Dosing complete in Phase 1/2a trial for injectable wet AMD therapy
➤ First patient dosed in light-activated optogenetic monotherapy study
➤ Phase 2 study for investigational new immunomodulator to treat dry eye disease
➤ Partnership created to develop tissue implant therapies
➤ ASCRS news and events
➤ 12-month Phase 1 trial data from sustained drug delivery option for anti-VEGF therapy
➤ Phase 1b results for possible topical treatment of corneal injuries
➤ Long-term safety and efficacy reinforced with 2-year clinical trial data of AMD therapy
➤ FDA accepts NDA for GA therapy
➤ NDA submitted for investigational dry eye treatment
➤ Company completes guidance meeting with FDA for dry eye drug
➤ Companies partner to conduct Phase 3 trials for possible NK treatment
➤ Partnership to develop gene therapy
➤ ASCRS news and events
➤ FDA 510(k) clearance granted to FLACS system
➤ New genetic eye disease discovered
➤ Primary endpoint met in Phase 3 trial for dry eye drug candidate
➤ New analysis of thyroid eye disease therapy
➤ Licensing agreement for commercialization of investigational myopia therapeutic
➤ ASCRS news and events
➤ Topline results from Phase 3 pivotal trial evaluating mydriasis drug
➤ First person dosed in Phase 3 trial for first-in-class dry eye candidate
➤ Phase 2 study of a novel glaucoma drop targeting non-traditional outflow
➤ Digital vision screening platform with machine learning
➤ Alcon to acquire dry eye drop and inflammation/pain drop
➤ New issue online
➤ ASCRS news and events
➤ Phase 4 results evaluating intravitreal implant
➤ Interim results for X-linked retinitis pigmentosa treatment
➤ New non-contact glaucoma laser treatment receives CE mark
➤ Companies collaborate for regenerative dry eye therapy
➤ Complimentary MIGS video series
➤ ASCRS news and events