The robust NK and PCED pharmaceutical pipeline
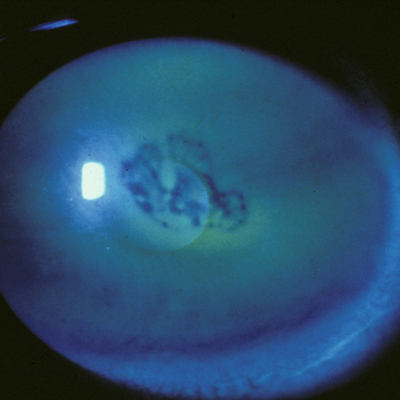
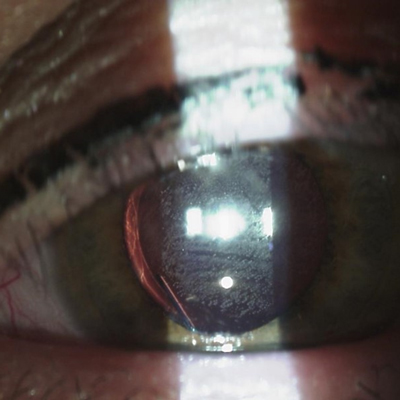
For about 7 years, there has been one FDA approved treatment for neurotrophic keratitis (NK): Oxervate (cenegermin-bkbj, Dompe). Before that, there was nothing. Now pharmaceutical options for NK—and persistent epithelial corneal defect (PCED) more generally—in the pipeline number about a half dozen.