Cornea: Changing mindsets
Summer 2025
by Ellen Stodola
Editorial Co-Director
Results from the Zoster Eye Disease Study (ZEDS) were first presented last year. This randomized clinical trial was conducted in 95 sites from November 2017 to June 2024, looking at the use of low dose valacyclovir for reducing complications with herpes zoster ophthalmicus (HZO). Elisabeth Cohen, MD, and Bennie H. Jeng, MD, spoke to EyeWorld about the results, highlighting recently published papers on findings from the study.
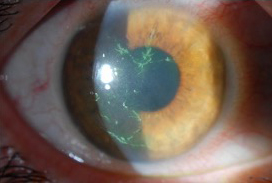
Source: Christopher J. Rapuano, MD
Since the 90s, we’ve learned that complications of zoster are associated with active varicella zoster virus infection, Dr. Cohen said. She noted the Herpetic Eye Disease Study, which looked at herpes simplex virus eye disease, showed that low-dose, prolonged acyclovir was effective in reducing recurrent episodes of disease during the 12 months that patients were on it but not during the 6 months after they stopped taking the low-dose acyclovir.
“In about 2010, I had the idea to try treating zoster the way we treat herpes simplex eye disease,” she said. “I had zoster in my eye in 2008 and had a bumpy course, and after a year, I had lost vision to the point that I had to give up being a regular cornea doctor, seeing patients, because I didn’t have the depth perception to do microsurgery anymore. I was very well taken care of, and every time I got worse, they gave me another week of high-dose antiviral treatment. Then I had this light bulb go off and thought, ‘Why don’t we do prolonged low-dose treatment for HZO like we do for HSV?’”
Zoster Eye Disease Study (ZEDS)
Dr. Cohen began the undertaking of writing the grant for what would become the Zoster Eye Disease Study (ZEDS), which was submitted in September 2013 with 60 U.S. centers agreeing to participate. It took three tries before it was finally funded by the National Eye Institute of the National Institutes of Health in 2016, Dr. Cohen said, adding that a lot of cornea doctors who were going to be involved with the study started using the treatment, despite a lack of good evidence to support its use.
The concept was to enroll HZO patients who had involvement in their eye within the year prior to enrollment. They could have a history of a typical rash any time in the past, but they needed to have active keratitis or iritis within 1 year of entering the study.
Dr. Jeng noted that the study was conducted in 95 centers across the world, mostly in the U.S. but also in Canada and New Zealand. There were 527 patients enrolled, and they were randomized to 1,000 milligrams per day of valacyclovir double masked versus placebo for 1 year of treatment and 18 months of follow-up.
“It is important to note, even at the outset of the study in the statistical analysis plan, we identified four different strata that patients would be randomized into,” Dr. Jeng said. These strata were by age—with cutoffs of younger than 60 years or 60 years and older—and time since onset of disease at enrollment—less than 6 months or greater than 6 months—to define recent onset versus chronic disease. The purpose of that was that it was expected for disease manifestations to vary by age. “We also expected a greater benefit of treatment in the recent onset group,” he said. He noted that 460 completed the 18-month study, which included 1 year of treatment of either drug or placebo and 6 months of follow-up when patients were off the treatment.
“We did it by age because we thought half of the people would be under 60 years and half 60 years or older, and disease manifestations, especially the chronic pain syndrome of post-herpetic neuralgia, differ by the age of onset,” Dr. Cohen said. “We did it by disease duration because we hypothesized that it would be more effective in people with recent onset disease, but because there are a lot of people out there with chronic disease, we didn’t want to exclude them from the study.”
Results
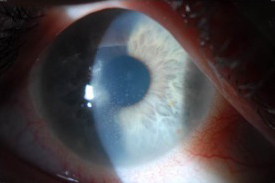
Source: Christopher J. Rapuano, MD
The primary endpoint was looking at whether there was a treatment benefit in reducing new or worsening corneal complications of stromal keratitis, endothelial keratitis, iritis, or dendriform epithelial keratitis at 12 months, while the secondary endpoint was looking at 18 months.1
With the first endpoint, the P value was 0.09, but the hazard ratio was 0.77, indicating a 23% reduction. At 18 months, the hazard ratio was 0.73, a 27% reduction, with a P value of 0.03. “We hit it at 18 months but missed on the primary endpoint,” Dr. Jeng said. When looking at the different strata, they didn’t find anything statistically significant. “We did find in one of the four strata, the less than 60-year-old recent onset disease group, the P value was 0.06, so very close. The hazard ratio was 0.63, a 37% reduction,” he said, which is clinically meaningful.
The hazard ratio was only a 4% difference at 12 and 18 months, Dr. Cohen said, but that 4% was the difference between statistically significant and not. “The good news is that we showed persistent benefit after the treatment was completed,” she said, adding that this is different than with herpes simplex. “That doesn’t surprise me in that with herpes zoster, there is a rocky course, then it quiets down, and one figures out the lowest dose of steroid to keep a person on so that they don’t flare up. But with herpes simplex, there are separate bouts of disease, separated often by years. So I think that the benefit at 18 months should determine treatment, and the data showed that the recent onset benefited more than the chronic disease.”
Dr. Cohen’s opinion is that if there is herpes zoster affecting the eye, after the patient finishes the 7–10 days of high-dose oral antiviral, they should be put on a year of low-dose valacyclovir as soon as possible.
Looking at post hoc analyses, Dr. Jeng said it was found that if you lumped all the recent onset patients in both age groups compared to chronic, there was a statistically significant reduction in endpoints at 1 year in the recent onset group that were treated with a P value of 0.03 and at 18 months with a P value of 0.02. “The other important secondary analysis that we found was that there was a statistically significant benefit with treatment in reducing subsequent endpoints,” he said. “Just because someone had an endpoint, we didn’t pull them out of the study; we kept them in their study group and continued to follow. We know that every endpoint can lead to more damage that can lead to vision loss, so we wanted to see if this treatment reduced subsequent endpoints.” There was a statistically significant P value of 0.02 both at 12 and 18 months if they were treated with valacyclovir in terms of reducing subsequent endpoints.
“We think the evidence supports suppressive valacyclovir treatment of 1,000 mg daily for 1 year to reduce these endpoints,” he said. “It was statistically significant at 18 months, and the recent onset patients were more likely to benefit than chronic disease patients. The evidence also showed that this treatment reduces multiple endpoints.”
Post-herpetic neuralgia
The secondary aim of the study was to look at post-herpetic neuralgia, and Dr. Jeng said this produced some interesting data.2 There were 73 participants in this group.
“What we found, in terms of prevalence, is that participants who were younger than 60 years had a significantly lower prevalence of post-herpetic neuralgia at 18 months. When we looked at pain scores, the younger patients with chronic disease had statistically significant lower pain scores at both 12 and 18 months. Those who were in the older chronic group had suggestion of decreased pain, but it wasn’t statistically significant,” he said.
Pain duration was examined as well. There was a statistically significant decrease in pain duration for those who were treated at 18 months. “What’s also interesting is there was a statistically significant reduction in the dose of neuropathic medications needed to be used for the patients who were on valacyclovir treatment,” Dr. Jeng said.
Showing that this treatment can reduce the amount of neuropathic pain medication needed was a benefit of the study, Dr. Cohen said, adding that this is important because the most common medicines that are used for pain related to zoster are pregabalin and gabapentin, and they have a lot of side effects. It’s not just for post-herpetic neuralgia, which is defined as pain beyond 3 months; Dr. Cohen thinks it may be valuable even earlier as neuropathic pain medications are also used for acute pain management.
ZEDS did not show any evidence of serious side effects with the low dose, though Dr. Cohen noted that it is a good idea to get a baseline test for kidney function as was done in the study. The low dose of valacyclovir being used is much better tolerated than neuropathic pain medicines, she added.
Further examinations and thoughts from the study
Dr. Jeng said the study also looked at the impact of vaccination against herpes zoster with the new vaccine that came out during the study and evaluated whether or not vaccinations against zoster affect COVID-19 diagnosis and severity.
In terms of the zoster vaccine, Dr. Cohen said the question was if a patient has zoster in the eye already, will the vaccine cause it to flare up. Patient aged 50 years and older should be getting the two-shot series of recombinant zoster vaccine, she said. For those who are immunocompromised, the CDC recommends that they get the two-shot series starting at age 19.
Dr. Jeng noted that the sample size for ZEDS was not exactly as hoped for, attributing this to the fact that a lot of cornea specialists thought before the trial results that this treatment reduced the endpoints; they already thought the treatment worked and prescribed it, so this made it harder to recruit patients. “The take-home message here is that we think that this treatment works,” he said. “If you ask me how’s it going to change my management of these patients, I would say that for individuals who have a history of zoster ophthalmicus and have any corneal findings, I would treat with 1 year of valacyclovir.”
Dr. Cohen said she would also like to encourage physicians to enter their patients into trials because it helps find out what works and what doesn’t. “Expert opinion is expert opinion, and data that you get with a randomized clinical trial is really the gold standard,” she said. “You want it to not only be effective, but you want it to be safe.” Dr. Cohen said there is a tremendous amount of data on valacyclovir. It is FDA approved at this dose and duration to reduce genital herpes simplex virus infections, and it’s been around for 30 years, she said, adding that it’s available in a generic version. “If it works, it’s safe, and it’s low cost, it’s something that we’d like to see used,” she said.
Article Sidebar
Changing mindsets

Regarding “Changing mindsets: current solutions and what’s ahead,” Rom Kandavel, MD, EyeWorld Cataract Editorial Board member, said, “The development of new MIGS devices makes me think that at this growth rate, in under 10 years we will be doing more glaucoma surgical procedures than cataract surgery.”
About the physicians
Elisabeth Cohen, MD
Professor and Vice Chair for Education, Faculty, and Academic Affairs
Department of Ophthalmology
NYU Grossman School of Medicine
NYU Langone Health
New York, New York
Bennie H. Jeng, MD
William F. Norris and George E. de Schweinitz Professor
Chair, Department of Ophthalmology
Director, Scheie Eye Institute
University of Pennsylvania Perelman School of Medicine
Philadelphia, Pennsylvania
References
- Cohen EJ, et al. Low-dose valacyclovir in herpes zoster ophthalmicus: The zoster eye disease randomized clinical trial. JAMA Ophthalmol. 2025;143:269–276.
- Warner DB, et al. Low-dose valacyclovir for postherpetic neuralgia in the Zoster Eye Disease Study: A randomized clinical trial. JAMA Ophthalmol. 2025;143:277–285.
Relevant disclosures
Cohen: None
Jeng: None
Contact
Cohen: Elisabeth.Cohen@nyulangone.org
Jeng: Bennie.Jeng@Pennmedicine.upenn.edu




