ONLINE EXCLUSIVE
Cornea
February 2023
by Ellen Stodola
Editorial Co-Director
I had the honor of co-founding The Winning Pitch Challenge with retina specialists John Pollack, MD, and David Williams, MD, in 2017. Our dream was to spur innovation by assisting ophthalmologists with great ideas looking for advice on whether their concept was commercially viable and, if so, help them with the necessary steps to maximize their chance for success. We thought that if we could improve and facilitate the pace of innovation, we would ultimately improve patient care in our world at a faster rate. The Winning Pitch Challenge website was designed to achieve this goal by providing ophthalmologists with free access to key educational resources, highly experienced mentors with relevant business and innovation cycle knowledge, networking opportunities, and exposure to potential financial resources and guidance that could help them overcome critical early-stage obstacles, such as securing intellectual property, developing a working version of the product, identifying a team, and designing early-stage studies. Our hope is that ophthalmologists with great ideas will engage and submit their ideas and ultimately, if chosen, present their idea in a Shark Tank-type setting at the Eyecelerator@ASCRS and Eyecelerator@AAO meetings.
The next Winning Pitch Challenge will take place Thursday, May 4, in San Diego, California, at Eyecelerator@ASCRS 2023. This will be a great session to attend for those interested in the innovation process to either submit their idea and potentially present or just come learn about the process and hear the three finalists’ innovation journeys as they pitch to four judges in an energy-filled session co-moderated by my partner John Berdahl, MD. This year, for the first time, we have a fifth judge—the audience! It is an exciting program, and the past winners have felt it helped them to either found their company or take their small company to another level on the world innovation stage.
—Vance Thompson, MD, Director of The Winning Pitch Challenge
Bala Ambati, MD, won The Winning Pitch Challenge during the session at the ASCRS Annual Meeting in 2019. His company, iVeena, presented the IVMED-80, a pharmacologic drop treatment for keratoconus.
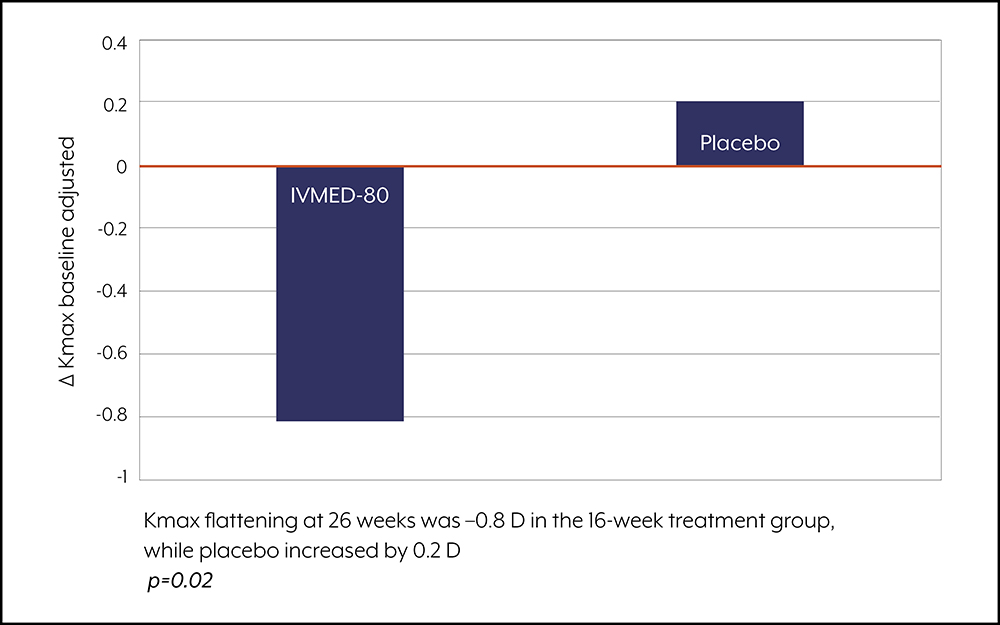
Since then, Dr. Ambati said a 6-month Phase 1/2a clinical study has been completed in 2021 led by Dr. Arturo Chayet. The study met the primary endpoint of a statistically significant reduction in baseline-adjusted Kmax of 1.0 D relative to placebo (p=0.0199; paired longitudinal analysis). In 2022, Glaukos and iVeena announced a strategic licensing agreement where Glaukos will develop and commercialize IVMED-80. As part of the agreement, Glaukos paid $10 million upfront and assumes costs of all development activities for IVMED-80. iVeena is eligible for development and sales milestones, as well as royalties.
Before winning The Winning Pitch Challenge, the initial experiments for the product took place in 2014. Glaukos’ first-generation corneal crosslinking therapy known as Photrexa, or epi-off, which is the only FDA-approved treatment shown to slow and halt the progression of keratoconus, involves debriding the top layer of the cornea. Dr. Ambati thought there should be a pharmacologic approach.
Source: iVeena
Through research, it was determined that copper was deficient in keratoconic corneas. It was also known that copper was a necessary cofactor for lysyl oxidase. There are genetic mutations in the lysyl oxidase gene family in these patients, he said, adding that Dr. Rohit Shetty in India has shown both from epithelial cells removed from keratoconic patients and SMILE lenticules in patients who went on to develop keratoconus that lysyl oxidase is reduced. “There’s evidence from multiple sources that lysyl oxidase deficiency is critical to the pathogenesis of keratoconus,” he said. “The experiment we did was: If we gave copper to corneal fibroblasts, the cells of the stroma, from normal cells versus keratoconic cells, what will happen?”
After doing that experiment, Dr. Ambati said that his laboratory team found that lysyl oxidase activity increased to normal levels in the keratoconic cells. This was the original scientific foundation of the iVeena drop product in 2014. “We did rabbit experiments for a few years and optimized the formulation, earned the orphan designation, and optimized the dosing regimen,” he said, adding that this put them in the position to start the Phase 1 clinical trial in early 2019, based on the results from the preclinical data.
Even though Glaukos has taken over the product, Dr. Ambati said he still expects to be involved. “We have a good working relationship with Glaukos,” he said. “They’re doing all the work now as far as the clinical process, but we will be in touch with them regularly.”
Dr. Ambati added that having a drop option for keratoconus could be complementary for patients in terms of a non-surgical option and potentially expand the pool of keratoconus patients who can be treated.
Dr. Ambati said iVeena is also investigating a pediatric myopia product, which he mentioned during his presentation at The Winning Pitch Challenge in 2019. “That’s our other product in the pipeline, so we’re devoting our resources to moving forward on a pediatric myopia clinical trial,” he said. The mechanism of action will be to stiffen the sclera by increasing lysyl oxidase. He added that there is already good animal data on this, and iVeena intends to initiate a clinical trial this year.
About the physician
Bala Ambati, MD
Pacific ClearVision Institute
Eugene, Oregon
Relevant disclosures
Ambati: iVeena
Contact
Ambati: bambati@pcvi.com