Cornea: Beyond the routine
Fall 2025
by Ellen Stodola
Editorial Co-Director
With many new cancer therapies in development, one emerging area is antibody drug conjugates. However, sometimes these therapies can pose a risk to the eye and cause ocular adverse events. Several experts spoke about the research in this space, what they’ve seen in terms of side effects, and considerations for patients taking these drugs and the eyecare providers monitoring them.
The ASCRS Cornea Clinical Committee recently hosted a webinar on the topic as well, “Antibody Drug Conjugates – An Emerging Class of Cancer Therapeutics Which Pose Risks to Corneal and Anterior Segment Health.” The webinar featured moderators Naveen Rao, MD, and Winston Chamberlain, MD, PhD, with speakers Kamran Riaz, MD, Stella Kim, MD, and Asim V. Farooq, MD. Speakers in the webinar noted the growing number of antibody drug conjugates (ADC) to discuss.
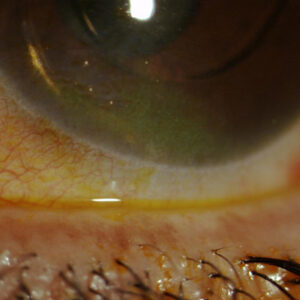
Source: Neel Pasricha, MD
Dr. Chamberlain explained during the webinar what an ADC is and how these therapies target cellular markers on tumors. Some of the major concerns with ADCs are that despite being thought of as a “magic bullet” because of their specificity, only a small percentage of the drug (a macromolecule) gets to its target.
Receptors on tumor cells may exist on healthy tissue as well, and this can produce a nonspecific uptake of the drug; there are many mechanisms by which this occurs. “But the end result is that it can lead to death or sickness of healthy cells, and we call these adverse events,” he said in the webinar.
One thing that may not be clearly understood is that when we kill tumor cells, we create bystander effects, he said. When cells die, they burst open and release immunological and inflammatory mediators and the toxic molecules bound to ADCs that can cause death, destruction, or sickness of neighboring cells. This can lead to bystander inflammation and destruction of healthy tissue that’s not targeted by an ADC.
ADCs have a different approach than traditional chemotherapy, said Neel Pasricha, MD. “Think of traditional chemotherapy as a toxic drug that’s given, and the cells that replicate the fastest, like the cancer cells, get damaged more than the healthy cells that aren’t replicating as fast,” he said. “But there are effects in healthy tissues because a lot of cells are replicating fast, like the skin and the hair.” This also includes the surface and epithelium of the cornea.
Dr. Pasricha said the first ADC approved in 2000 was a breakthrough. The rationale behind it was to take the chemo component, the toxic drug, and link it to an antibody, and the antibody would shuttle the chemotherapy to its target. “In theory, it sounds like a great idea, and it had great impact, but there’s still a lot of off-target effects from these targeted chemotherapies,” he said.
In the webinar, Dr. Chamberlain said there has been a burst in ADCs in the last decade. There are more than 200 ADCs being investigated or currently in clinical trials. “As ophthalmologists, we have to be aware of a large surge of drugs that are going to come forward that our patients may be on and be aware of potential effects on the ocular surface,” Dr. Chamberlain said. ADCs have many targets, including breast cancer and ovarian cancer. Many of the ADC therapies are not supposed to cluster in eye tissue, but many still affect the eye.
Dr. Pasricha said that around 50% of all ADCs have some degree of ocular toxicity. Around 10–20% of patients on these drugs will have mild ocular surface toxicity. For other drugs, the ocular surface toxicity is the number one adverse event. For example, he mentioned belantamab mafodotin, which is used to treat multiple myeloma; more than 70% of patients had corneal toxicity in clinical trial data. Real- world data suggests it could be even higher.
In addition to being the number one adverse event, it can also be the dose-limiting adverse event, preventing the patient from getting the cancer drug as frequently due to its impact on the ocular surface.
In the ASCRS webinar, faculty members highlighted their concerns about the number of ADCs, with Dr. Farooq commenting that with drugs currently in development, this could be a huge wave of patients. “In ophthalmology and optometry, we all have to be prepared and plan ahead for the number of patients who will be on these drugs,” he said. These are patients who often will require regular eye exams with a cadence that might be determined by regulatory policy, depending on toxicity.
Dr. Farooq discussed some of the diagnostics and how to define the nature of the disease. Sometimes the patient may not have a lot of symptoms other than blurred vision. A slit lamp exam with vital dye stain is important, and eyelid eversion can be helpful to get at the extent of ocular surface involvement. He also suggested using refraction and topography, when available, to help understand the impact on vision. Other things like confocal microscopy of the corneal epithelium or corneal nerves may be used.
Commenting further to EyeWorld, Dr. Farooq said, “I think one of the more common signs is what I have called pseudomicrocysts. These are cystic-appearing lesions in the epithelium of the cornea.” These pseudomicrocysts generally start in the periphery of the cornea, and with time and subsequent doses, they migrate toward the center.
Having seen these patients for several years, we know the lesions tend to go away if the patient is on a dose hold, dose reduction, or the drug is stopped, he said. However, these pseudomicrocysts are impacting vision. Patients can have changes in vision, including from refractive shift.
Patients can also experience glare, light sensitivity, or irritation, and some of that may be related to the lesions and what they’re doing in terms of light scatter or from other mechanisms.
We also have anecdotally seen worsening or onset of dry eye, indicated by punctate staining of the corneal epithelium with fluorescein, Dr. Farooq said. Some patients have increased staining of the cornea. In some cases, there can be a whorl-like pattern of staining with fluorescein. This is separate from the pseudomicrocysts or the general pattern of punctate corneal staining. This whorl pattern seems to start from the corneal periphery. This could indicate a form of limbal stem cell dysfunction. These findings tend to improve with dose modifications and supportive treatments.
Dr. Pasricha also noted patients presenting with corneal pseudomicrocysts. Initially patients are asymptomatic. Soon after they develop these, they develop symptoms and start to get breakdown of the epithelium.
Additionally, he said it’s been found that the pseudomicrocysts cause a refractive change in the cornea. “When they start off in the periphery, they’re steepening the periphery, so it’s causing a hyperopic shift. As they move to the center, they’re steepening the center, so it’s a myopic shift,” he said. “For patients, it’s frustrating because it’s like having a moving target.”
Fortunately, these changes so far are reversible. If you stop the ADC, things will get better over time, he said. “What’s interesting is if it was truly just affecting the epithelium and going away, one would expect within a week or two that it would be better because the corneal epithelium turns over about every week. But we’ve found it takes anywhere from 2–6 months for it to fully reverse, and I’ve even had some patients where it takes a year to get fully back to normal. These ADCs are thought to accumulate in the basal epithelium and stay there like a reservoir.”
These issues are something that companies working on ADCs are aware of, Dr. Pasricha said, and ophthalmologists and oncologists are becoming more aware as well. The problem, he said, is that the pre-clinical models to look at these toxicities are not great. Even when doing animal studies, they don’t always find the same corneal problems. Companies are spending a lot of time and money to develop these drugs, doing cell culture safety, and animal safety models, and they find a reasonable safety profile pre-clinically, but then they sometimes learn about issues in the Phase 1 trial. That’s a huge limitation.
The holy grail would be to design ADCs that don’t have this toxicity, Dr. Pasricha said, adding that there are 11 FDA-approved ADCs, and three have a black box warning from the FDA, which is the highest warning they can issue about a drug toxicity. With so many ADCs in clinical trials, Dr. Pasricha said this will likely be a big issue for at least the next 10–20 years.
While he said it doesn’t seem to be one type of antibody that causes this toxicity, there is research being done into what makes an ADC more likely to cause toxicity. It could be something with charge distribution or hydrophobicity impacting how easily it can cross the cell membrane. The general hypothesis of how this is happening is the process of macropinocytosis, which means the cell membrane remodels and “drinks” things non-specifically from the extracellular fluid, he said. This off-target nature makes it a hard problem to fix.
Dr. Pasricha has recently been a part of an effort to form a multicenter consensus on grading of these ocular toxicity issues. The idea for this started when he was speaking with Laura Esserman, MD, head of the University of California, San Francisco breast cancer center. She had asked for Dr. Pasricha’s help looking at the corneal epithelium in patients in trials for breast cancer drugs.
“When I started seeing these patients, one of the first problems I had was they would come to my clinic with ocular toxicities, and the grading scale was difficult for me to understand,” he said. “They had confusing terms. I had to grade their dry eye score, their keratitis score, their blurred vision score. I had all of these categories to grade the same underlying problem.” This set Dr. Pasricha on the path to making a better grading scale. Dr. Esserman suggested that he consult with ophthalmologists, oncologists, and FDA representatives to tackle the issue.
“It wasn’t to replace the gold standard, which is the Common Terminology Criteria for Adverse Events (CTCAE), but it was to have a better consensus scale that would make it easier to speak the same language,” Dr. Pasricha said.
Some of the major advances from this effort were separating out the visual acuity from the objective clinical findings. We didn’t include a vision scale in the cornea score, he said. It was a separate visual acuity score and cornea score. “We separated the signs and symptoms, we had representative pictures, and we had recommended changes to the oncology drug,” he said.
Dr. Farooq and Dr. Chamberlain worked with Dr. Pasricha and others in this effort. The CTCAE has historically been used across oncology trials. “This space of targeted cancer drugs and ocular adverse events is growing, and there’s some unique findings and considerations, so we thought the CTCAE wasn’t adequate for facilitating clear communication between ophthalmologists and oncologists.”
He added that many of these clinical trials and different drugs had their own system and scale. “The idea was to get some minds together who have different perspectives and come up with a grading scale that could be used across different targeted cancer drugs.
“Originally, this was thought to be just for ADCs, but once we started getting this panel together, we decided we should make it relevant for any experimental oncology drug,” Dr. Pasricha said. “For each grade, there’s an oncology drug recommendation.” The work done by this group is currently under review with JAMA Ophthalmology.
Stopping the drug, reducing, or delaying the dose does help, he said. It’s important to provide some guidance on when you should dose delay, dose reduce, or discontinue the drug. However, the guidance can’t be so stringent that every grade 3 needs to be stopped. “We tried to build things like that into the guidelines, where there’s flexibility in the framework to account for unique patient clinical scenarios,” he said.
For most oncologists, when a patient complains of eye pain or blurry vision, they’ll refer to the eyecare provider, and the same with the eyecare provider—when we see toxicities in the eye, we aren’t comfortable saying that they should stop a drug. It is so important to have team communication, he said.
We need to carry this throughout the country, Dr. Pasricha said, noting that ADCs are given at infusion centers. It’s important to have an eyecare provider as a regional champion about ocular toxicities with these, he said.
“I think discussions like this are important because the more people working on this, the faster we’ll find better solutions,” Dr. Pasricha said. His lab is working on developing eye drops to address this. Right now, there’s no known effective therapy except dose interruption. There’s a huge need for an effective therapy that will allow patients to be on the drug without as many side effects or be on a higher dose without the eye problems, he said.
About the physicians
Winston Chamberlain, MD, PhD
Affiliate Professor of Ophthalmology
Casey Eye Institute
Oregon Health & Science University
Northwest Permanente
Portland, Oregon
Asim V. Farooq, MD
Professor of Ophthalmology and Visual Science
University of Chicago
Medical Center
Chicago, Illinois
Neel Pasricha, MD
Assistant Professor of Ophthalmology
University of California, San Francisco
Francis I. Proctor Foundation
San Francisco, California
Relevant disclosures
Chamberlain: AbbVie, GlaxoSmithKline
Farooq: AbbVie, Ambrx, Amgen, AstraZeneca, Boehringer Ingelheim, Eisai, GlaxoSmithKline, Mythic Therapeutics, Pfizer, Sanofi, Santen, Skye Bioscience, Vicore Pharma
Pasricha: AbbVie, Amgen, AstraZeneca, Sanofi
Contact
Chamberlain: chamberw@ohsu.edu
Farooq: afarooq@bsd.uchicago.edu
Pasricha: Neel.Pasricha@ucsf.edu