Cornea
July 2021
by Liz Hillman
Editorial Co-Director
Keratoprosthesis (KPro) is not frequently discussed in the greater ophthalmic community. It is a rare procedure and there is only one device approved by the FDA for use in the U.S., the Boston KPro. Since its initial FDA approval in 1992, less than 15,000 Boston KPro procedures have taken place as of 2019.1
Keratoprosthesis is an active area of research and development, however, and Sophie Deng, MD, PhD, said there have been recent innovations and updates to some devices in the keratoprosthesis field as a whole.
Boston KPro update and insights
The Boston KPro is the most commonly referenced keratoprosthesis in the U.S. In general, there have not been major changes to the Boston Type I KPro within the last 5 years, Dr. Deng said. Updates from the original design that occurred prior were the option for a titanium backplate that was slightly larger than the original PMMA backplate (the latter of which is still available, and Dr. Deng uses PMMA) and a snap-on backplate design (the previous version was screw on).
Boston KPro allows for a quick visual recovery and excellent visual outcome shortly after surgery, Dr. Deng said. The key for long-term success with KPro is in the postop regimen, she said. It is significantly more labor intensive than a corneal transplant, with patients on anti- inflammatories, antibiotics, antifungals, and sometimes glaucoma medications. She noted that it’s now standard of care for a patient with glaucoma to have a combined Boston KPro and tube shunt procedure.
Weekly follow-up continues until the corneal surface is re-epithelialized, Dr. Deng said. After the patient is stable, the follow-up interval is adjusted to usually every 2–4 months.
Dr. Deng said ophthalmologists performing follow-up care locally for KPro patients need to be familiar with all the possible complications associated with this procedure.
“We have seen patients become NLP because the local ophthalmologists were not familiar with the risk of glaucoma. This is the leading cause of loss of vision in long-term follow-up. The other is infection. The infection is devastating and can blind people,” she said.
“The ophthalmologist needs to recognize if there is any infiltrate in the donor tissue adjacent to the KPro and any epithelial defect or anterior chamber reaction that could indicate there is infection in the cornea. If they see those signs, the patients need to be treated with more aggressive antibiotic regimen. After keratoprosthesis, the patients will be on antibiotics for the rest of their life,” Dr. Deng said. “The most common pathogen of infection is microbial followed by fungi. Fungal infection is more difficult to eradicate.”
A newcomer in the KPro field
There are many players in the KPro field available globally, some with more nuanced indications and/or different designs and materials. Earlier this year, the CorNeat KPro (CorNeat Vision), a newcomer to the space, made headlines.
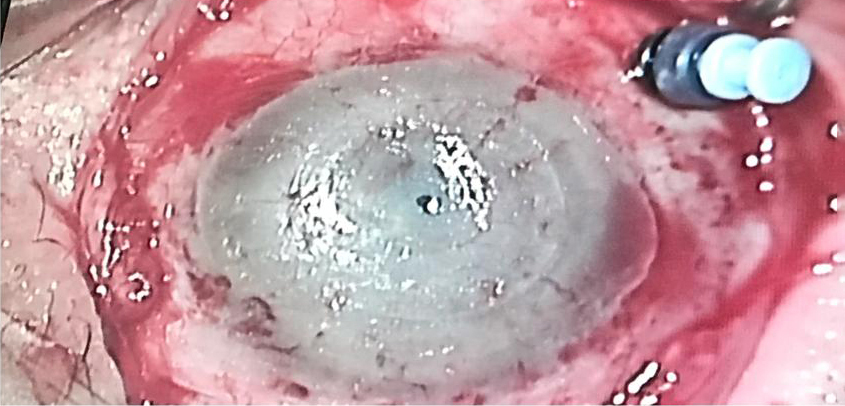
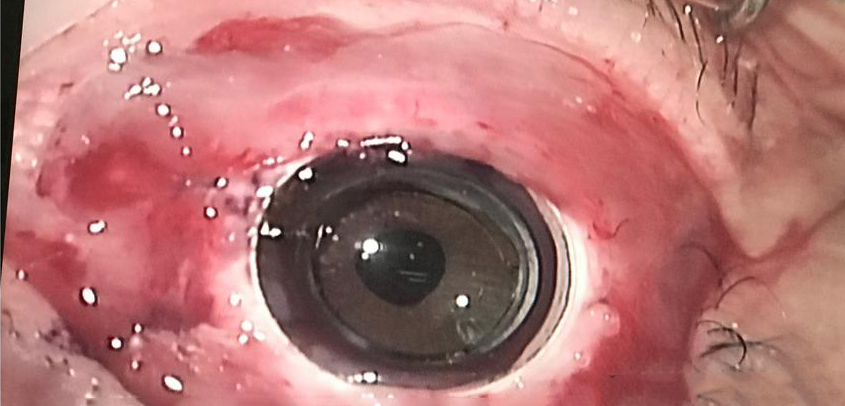
Source: Gilad Litvin, MD
The CorNeat KPro is described on the company’s website as a “patented synthetic cornea utilizing advanced cell technology to integrate artificial optics within resident ocular tissue. It is produced using nanoscale chemical engineering, which stimulates cellular growth.” The CorNeat KPro is designed to integrate under the conjunctiva. Surgically, the CorNeat KPro implantation procedure is designed to reduce the time of “open sky.”
Irit Bahar, MD, MHA, who performed the first-in-human CorNeat KPro procedure in January 2021, said the surgical sophistication needed for most KPro procedures and the high rate of complications, due to the procedure or materials, motivated creation of the CorNeat KPro. Prof. Bahar said the first patient who received the CorNeat KPro has been having weekly follow-up with the results exceeding expectations thus far.
Gilad Litvin, MD, creator of the CorNeat KPro, noted that the synthetic nature of the device makes timing of the procedure more flexible, compared to those reliant on donor tissue.
“Once you don’t need an organic tissue, you can time the implantation at the surgeon’s and patient’s convenience,” he said.
“The attachment site of the CorNeat KPro is external to the eye, thus no interference with ocular physiology occurs,” Dr. Litvin explained. The device is also designed to allow for future anterior segment procedures.
“Follow-up is expected to be much simpler than previous [KPro] models. There is no need for protective contact lenses and no need for lifelong, local antibiotic treatment,” Dr. Litvin said.
Dr. Deng, who said she only has hands-on experience with Boston KPro technologies, shared some thoughts on innovations like the CorNeat KPro. She said newer KPro technologies try to reduce risk of rejection with biointegratable materials, while also eliminating reliance on donor corneal tissue.
“Other options are being developed to try to solve the problem of integration and stability and the need for donor tissue,” she said.
Ophthalmologists’ daily work is vision preservation and restoration, but the KPro cases, Prof. Bahar said, are particularly emotional.
“This process requires both physical and mental resources, which few centers in the world can afford,” she said. “The KPro surgery instills hope for restoring the patient’s vision. The patient, his family, and the medical staff treating him are all emotionally involved. When we succeed, it’s amazing and it’s a supreme feeling of fulfilling our mission as ophthalmologists, but when it does not materialize or if it gets complicated, the frustration is equally great for everyone involved.”
ARTICLE SIDEBAR
Different KPro devices*
Boston KPro Type I (developed at Massachusetts Eye and Ear Infirmary): FDA approved; indicated for patients with multiple corneal graft failures, Stevens-Johnson syndrome, ocular cicatricial pemphigoid, and more2
Boston KPro Type II: FDA approved; similar to Boston KPro Type I but indicated for “severe end-stage ocular surface disease desiccation”2
CorNeat KPro: Similar indications as the Boston KPro; no donor corneal tissue used; made of synthetic, biointegratable material
Modified Osteo-Odonto-Keratoprosthesis (OOKP): Indicated for bilateral end-stage ocular surface disease, autoimmune diseases, and chemical injury; includes a “biological haptic” made from the patient’s tooth and alveolar bone3
Miro Cornea UR (Miro Vision): A synthetic, “bio-coated” keratoprosthesis that can support epicorneal or intrastromal implantation, according to the company’s website;4 indicated for eyes bilaterally blind from corneal disease or injury
Auro KPro (Aurolab): Described on the company’s website as similar to the Boston KPro in design and indicated for eyes with opacified corneas that have failed corneal grafts
LVP KPro: Described as an alternative MOOKP for eyes that are completely dry with end stage corneal blindness5
*This is not an exhaustive list, and some devices might not be available in the U.S.
About the physicians
Irit Bahar, MD, MHA
Director
Department of Ophthalmology
Rabin Medical Center
Petah Tikva, Israel
Sophie Deng, MD, PhD
Joan and Jerome Snyder Chair in Cornea Diseases
Stein Eye Institute
University of California, Los Angeles
Los Angeles, California
Gilad Litvin, MD
Chief Medical Officer
CorNeat Vision
Ra’anana, Israel
References
- Chodosh J. FDA approval obtained for the Boston Keratoprosthesis Type 1 Lucia Design. Boston KPro News. July 2019.
- Klufas M, et al. Boston Keratoprosthesis (KPro). eyewiki.aao.org/Boston_Keratoprosthesis_(KPro). Accessed Mar 3, 2021.
- Ortiz-Morales G, et al. Modified Osteo-Odonto-Keratoprosthesis. eyewiki.aao.org/Modified_Osteo-Odonto-Keratoprosthesis. Accessed Mar 3, 2021.
- Miro Cornea UR. www.Miro-Vision.de. Accessed Mar 3, 2021.
- Basu S, et al. LVP keratoprosthesis: anatomical and functional outcomes in bilateral end-stage corneal blindness. Br J Ophthalmol. 2019;103:592–598.
Relevant disclosures
Bahar: None
Deng: None
Litvin: CorNeat Vision
Contact
Bahar: iritbahar@gmail.com
Deng: deng@jsei.ucla.edu
Litvin: gilad@corneat.com